ABSTRACT
Vitamin B12 (cobalamin) is an essential water-soluble micronutrient and a critical cofactor in one-carbon metabolism, acting synergistically with folate to support DNA synthesis, methylation, and nervous system integrity. Deficiency is highly prevalent worldwide, particularly among pregnant women and women of reproductive age in low- and middle-income countries. Growing evidence identifies inadequate maternal B12 status as a major risk factor for a spectrum of adverse reproductive and perinatal outcomes, including female infertility, recurrent pregnancy loss, preterm birth, low birth weight, neural tube defects, and impaired fetal neurodevelopment. Mechanistically, B12 deficiency disrupts the methionine cycle, leading to hyperhomocysteinemia, which compromises oocyte quality, implantation, placental development, and epigenetic programming of the fetus. Maternal B12 insufficiency may also predispose offspring to long-term metabolic and neurocognitive sequelae, consistent with the Developmental Origins of Health and Disease framework. This narrative review synthesizes evidence from observational studies, randomized controlled trials, and mechanistic research, linking maternal B12 status to preconception health, assisted reproductive technology (ART) outcomes, adverse pregnancy events, and long-term neurodevelopment in offspring. We highlight the urgent need for targeted preconception and early antenatal screening, particularly using functional biomarkers such as holotranscobalamin, alongside evidence-based supplementation strategies. Implementing these interventions in high-risk populations represents a cost-effective and high-impact approach to reduce preventable maternal, fetal, and neonatal morbidity associated with early-life B12 deficiency.
Keywords: Vitamin B12; Cobalamin; Pregnancy; Neural tube defects; Preterm birth; Reproduction; Infertility; Maternal nutrition.
INTRODUCTION
The Critical Role of Cobalamin in Reproduction
Vitamin B12 (cobalamin) is an essential water-soluble vitamin required for the remethylation of homocysteine to methionine, a crucial biochemical reaction that underpins DNA synthesis, methylation processes, and the broader landscape of epigenetic regulation. When B12 levels fall below physiological needs, these fundamental one-carbon metabolic pathways become disrupted, leading to elevated homocysteine (Hcyt), impaired nucleotide synthesis, and widespread alterations in cellular function. Although the hematologic manifestations-such as megaloblastic anemia-and the neuropsychiatric consequences of deficiency are well established in clinical practice, its implications for reproductive health are increasingly evident and alarming [1].
Maternal B12 deficiency has the potential to alter several stages of reproductive physiology, including gametogenesis, oocyte maturation, and the earliest steps of embryogenesis. Elevated homocysteine, commonly associated with low B12 status, has been mechanistically linked to recurrent pregnancy loss [2], placental dysfunction, and a spectrum of adverse pregnancy outcomes. Low maternal B12 during the periconceptional (PCT) period and pregnancy is strongly associated with neural tube defects (NTD) [3], preterm birth, low birth weight, and impaired fetal growth, while exposed offspring may face altered neurodevelopment and long-term metabolic programming. These risks are of particular concern in populations whose dietary patterns involve minimal intake of animal-derived foods-such as vegans or strict vegetarians-highlighting an important and persistent public health issue.
The global burden of maternal B12 deficiency is substantial, representing a significant challenge in maternal and child health. Prevalence estimates suggest that more than 50% of pregnant women in certain high-risk regions, including South Asia, are affected [4]. Mechanistically, the B12-dependent methionine cycle produces S-adenosylmethionine (SAM), the universal methyl donor indispensable for proper epigenetic programming. Hence, inadequate B12 compromises the delicate balance of methylation required for successful implantation, rapid placental development, and subsequent organ formation [5]. This disruption is particularly consequential given the rapid cellular proliferation and heightened demand for one-carbon substrates during critical periods of fetal development.
This comprehensive narrative review draws together evidence from observational research, supplementation trials, and mechanistic studies to clarify the essential relationship between maternal B12 status and reproductive, pregnancy, and neonatal outcomes. It seeks to map current knowledge, identify key gaps that warrant further investigation, and support the development of targeted, evidence-based intervention strategies.
Research Strategy and Study Selection
This narrative review was conducted using a structured, non-systematic search strategy designed to synthesize current evidence on maternal vitamin B12 status and reproductive outcomes. Electronic databases including PubMed/MEDLINE, Embase, and Web of Science were searched for articles published between January 2000 and December 2025.
The following keywords and Medical Subject Headings (MeSH) terms were used in various combinations: “Vitamin B12” OR “cobalamin” AND “pregnancy,” “infertility,” “recurrent pregnancy loss,” “early pregnancy loss,” “neural tube defects,” “preterm birth,” “low birth weight,” “gestational diabetes,” “homocysteine,” “one-carbon metabolism,” “epigenetics,” “MASLD,” “metabolic dysfunction-associated steatotic liver disease,” and “maternal nutrition.”
Priority was given to systematic reviews, meta-analyses, randomized controlled trials, large prospective cohort studies, and mechanistic experimental research. Reference lists of selected articles were manually screened to identify additional relevant publications. Studies not available in English and those lacking primary data were excluded unless highly relevant for mechanistic context.
This approach aimed to provide a comprehensive synthesis of epidemiological, clinical, and biological evidence while identifying areas requiring further investigation.
Maternal Vitamin B12 Deficiency and Adverse Reproductive and Pregnancy Outcomes
Infertility and Recurrent Pregnancy Loss
Maternal vitamin B12 deficiency has been consistently implicated in impaired reproductive function, with strong evidence linking inadequate cobalamin status to both infertility and recurrent pregnancy loss (RPL) (Table 1). In a seminal case series, Molloy et al. (2001) reported substantial reproductive morbidity among women with confirmed B12 deficiency: across 15 reproductive attempts, 11 episodes of recurrent pregnancy loss and 4 cases of prolonged infertility were observed [3]. These findings underscore the significant reproductive burden associated with deficient one-carbon metabolism [6]. Vitamin B12 deficiency disrupts methylation pathways, leading to hyper Hcyt, which negatively affects endothelial function, promotes hypercoagulability, impairs implantation, alters ovulatory function, and reduces oocyte quality.
Quantitative analyses further illustrate the impact of B12 status on oocyte competence. Women with follicular fluid B12 concentrations >500 pg/mL were 2.3 times more likely to produce high-quality embryos than those with lower levels [8]. Similarly, in cohorts undergoing Assisted Reproductive Technologies (ART), women in the highest quartile of serum B12 (>701 pg/mL) exhibited a 2.04-fold higher probability of achieving live birth compared with those in the lowest quartile [7]. These data indicate that maintaining high-normal B12 concentrations during the PCT period is critical for optimal one-carbon flux, early embryogenesis, and placental development.
Vitamin B12 interacts with folate (vitamin B9) in methylation-dependent processes. While folate is well-recognized for NTD prevention, insufficient maternal B12 independently increases the risk of NTDs [3], preterm delivery, and neonatal deficiency, particularly when fetal stores are low or breastmilk B12 content is insufficient. Despite these implications, low maternal B12 remains under-studied, highlighting the need for targeted research to inform screening and intervention strategies.
This comprehensive narrative review draws together evidence from observational research, supplementation trials, and mechanistic studies to clarify the essential relationship between maternal B12 status and reproductive, pregnancy, and neonatal outcomes [3, 9]. It seeks to map current knowledge, identify key gaps that warrant further investigation, and support the development of targeted, evidence-based intervention strategies.
Early Pregnancy Loss and Hyperhomocysteinemia
Low maternal B12 and resultant hyper Hcyt are increasingly recognized as major risk factors for early pregnancy loss (EPL). A meta-analysis by Bala et al. (2021) demonstrated that the combined presence of low B12 and elevated homocysteine conferred a pooled OR of 4.98 for EPL [10]. Supporting this, a case-control study of 83 women with confirmed EPL reported that low B12 (P < 0.01) and elevated homocysteine (P < 0.05) independently predicted pregnancy loss, whereas folate deficiency was not significant. The combination of B12 deficiency and hyper Hcyt produced the highest risk (OR = 4.98; P = 0.002) [10].
Mechanistically, hyper Hcyt exerts endothelial and trophoblastic toxicity, impairs spiral artery remodeling, and compromises placental vascularization, while B12 deficiency reduces methyl-group availability, impairing DNA synthesis, methylation-dependent gene regulation, and trophoblast proliferation [11]. These convergent pathways provide a biologically plausible explanation for the observed EPL risk.
Clinically, these data support the integration of serum B12 and homocysteine measurement into the evaluation of women with recurrent or unexplained EPL. Early detection and correction may reduce recurrence risk and improve pregnancy outcomes (Table 1).
Neural Tube Defects and Major Congenital Anomalies
Low maternal B12 is robustly associated with neural tube defects (NTDs) and other congenital anomalies. Vitamin B12, alongside folate, is essential for one-carbon metabolism, methylation reactions, and DNA synthesis, all critical for neural tube closure during early embryogenesis [12]. A 2019 meta-analysis of multiple international cohorts demonstrated that maternal B12 deficiency significantly increased NTD risk (OR = 1.95; 95% CI: 1.38-2.76), independently of folate status [4, 13]. This confirms that B12 deficiency is a distinct and modifiable risk factor rather than a passive cofactor in folate metabolism.
Beyond NTDs, maternal low B12 is associated with other congenital anomalies. A 2020 meta-analysis reported an increased risk of congenital heart defects (OR = 1.34; 95% CI: 1.13-1.58) [13]. Collectively, these findings underscore the importance of monitoring maternal B12 status and considering combined supplementation with B12 and folic acid for optimal PCT prevention strategies.
Preterm Birth, Low Birth Weight, and Long-Term Offspring Outcomes
Maternal B12 deficiency is strongly linked to adverse perinatal outcomes, including preterm birth (PTB) and low birth weight (LBW) (Table 1) [14]. A systematic review and individual participant data meta-analysis (IPD-MA) incorporating 46 cohort studies identified serum B12 <148 pmol/L as an independent risk factor [5, 8]. Deficiency increased LBW risk by 15% and PTB by 21% (OR = 1.21; 95% CI: 1.09-1.34 for PTB) [15, 16]. The global prevalence of maternal B12 deficiency remains high, affecting 40-70% of pregnant women in regions such as India [17].
Mechanistically, adverse outcomes are mediated by hyper Hcyt and placental dysfunction, impairing trophoblast proliferation and nutrient transport. Long-term consequences for offspring include impaired cognition, increased adiposity, and insulin resistance. Seven RCTs have demonstrated benefits of maternal B12 supplementation on neurocognitive outcomes, though effects on birth weight remain variable [18]. High-quality evidence supports B12 deficiency as a risk factor for NTDs and LBW, with moderate-quality evidence linking it to gestational diabetes and long-term offspring metabolic health.
Early detection and supplementation of maternal B12 during preconception and pregnancy are therefore critical to mitigate perinatal morbidity and support optimal neurodevelopment and metabolic outcomes [19].
Maternal B12 Status and Gestational Diabetes Mellitus
The relationship between maternal B12 status and gestational diabetes mellitus (GDM) remains incompletely defined. Mechanistically, B12 serves as a cofactor in fatty acid and energy metabolism, implicating it in insulin sensitivity and GDM pathogenesis. Deficiency may impair methylation-dependent regulation of gene expression and mitochondrial function, theoretically predisposing to altered glucose homeostasis [3, 20].
A recent systematic review and meta-analysis, including 12 studies, found that serum B12 concentrations were lower in women with GDM compared with normoglycemic controls, but no statistically significant overall association with GDM risk was observed [20]. Qualitative analyses showed heterogeneity: four of six studies reported that higher or normal B12 levels were associated with reduced GDM risk, while the remaining studies found no effect. Serum folate levels were generally not associated with GDM, though higher red blood cell (RBC) folate-a marker of long-term folate status-was linked to increased GDM risk in some cohorts.
These findings highlight the complexity and conflicting nature of current evidence, emphasizing the need for large-scale, multi-regional prospective studies. Future research should also assess whether specific B12 metabolites, such as holotranscobalamin (HoloTC), or functional ratios with folate and homocysteine, provide better predictive value for GDM risk and could inform targeted interventions.
|
Category |
Outcome / Finding |
Evidence / Study |
Notes / Implications |
|
Infertility & Recurrent Pregnancy Loss |
B12 deficiency linked to infertility and recurrent fetal loss |
Molloy et al. (2001): 14 women, 15 reproductive episodes (11 losses, 4 prolonged infertility) |
Elevated homocysteine may impair implantation, ovulation, and oocyte quality. Folate deficiency historically linked to megaloblastic anemia. |
|
Early Pregnancy Loss (Miscarriage) |
Low B12 & high homocysteine increase EPL risk |
Bala et al. (2021): Case-control study (83 EPL, 70 controls); meta-analysis OR = 4.98 |
Folate deficiency not significantly associated with EPL. Screening B12 & homocysteine may help identify at-risk women. |
|
Neural Tube Defects (NTDs) & Congenital Anomalies |
Low maternal B12 associated with higher NTD risk |
Nie et al. (2025): 38 studies, 2,316 NTD cases; SMD = -0.23 |
Risk persists even with normal folate; combined B12 + folic acid supplementation recommended. |
|
Preterm Birth & Low Birth Weight |
B12 deficiency (<148 pmol/L) increases risk |
Systematic review & meta-analysis: 46 studies included |
Low B12 associated with preterm birth (+21%) and low birth weight (+15%). High prevalence in India (40-70%). Offspring long-term risks include impaired cognition, insulin resistance. |
|
Gestational Diabetes Mellitus (GDM) |
Evidence conflicting |
Wang et al. (2021): 12 studies; lower B12 in GDM but no consistent association |
RBC folate may increase GDM risk; overall, relationship unclear-more large-scale studies needed. |
Table 1: Impact of Maternal Vitamin B12 and Folate Status on Reproduction and Pregnancy Outcomes
Mechanistic Insights
Classically Recognized Mechanisms Linking B12 Deficiency to Adverse Pregnancy Outcomes
Vitamin B12 deficiency disrupts interconnected biochemical, epigenetic, and cellular pathways that collectively compromise maternal-fetal health. A primary consequence is impaired remethylation of homocysteine to methionine, resulting in elevated homocysteine levels that contribute to endothelial dysfunction, oxidative stress, and a prothrombotic state. These vascular effects can impair placental perfusion, ultimately restricting oxygen and nutrient delivery to the developing fetus and increasing the risk of fetal growth restriction, preterm birth, and low birth weight [3, 21, 22].
In parallel, insufficient B12 compromises DNA synthesis, disproportionately affecting rapidly dividing cells such as trophoblasts and embryonic stem cells, both essential for implantation, placental development, and early organogenesis. This impairment increases susceptibility to neural tube defects (NTDs) and other congenital anomalies, as demonstrated in human and experimental studies [5, 23, 24].
Vitamin B12 deficiency also reduces the production of S-adenosylmethionine (SAM), the universal methyl donor required for DNA and histone methylation. Lower SAM availability alters global and gene-specific epigenetic patterns in the placenta, affecting pathways involved in angiogenesis, hormone biosynthesis, and nutrient transport [5]. These epigenetic alterations link maternal micronutrient status to both immediate pregnancy outcomes and long-term offspring health.
Evidence from animal and experimental models reinforces these mechanisms: maternal B12 deficiency leads to reduced placental weight, restricted fetal growth, and persistent metabolic changes in offspring. Such findings align with the Developmental Origins of Health and Disease (DOHaD) framework, suggesting that periconceptional and gestational B12 status can program long-term susceptibility to adiposity, insulin resistance, and metabolic syndrome [25]. Clinically, this mechanistic cascade supports observed associations between maternal B12 deficiency and infertility, early pregnancy loss, NTDs, preterm birth, low birth weight, and later-life metabolic disorders [21, 22].
Finally, interactions between B12, folate, and homocysteine further emphasize the need for adequate one-carbon metabolism. Reduced methylation capacity may exacerbate folate-related disturbances in nucleotide synthesis and DNA repair, highlighting the combined importance of these micronutrients for optimal placental and fetal development. Together, these mechanistic insights provide a strong rationale for periconceptional B12 supplementation to promote healthy pregnancy outcomes and mitigate adverse long-term metabolic programming (Figure 1).
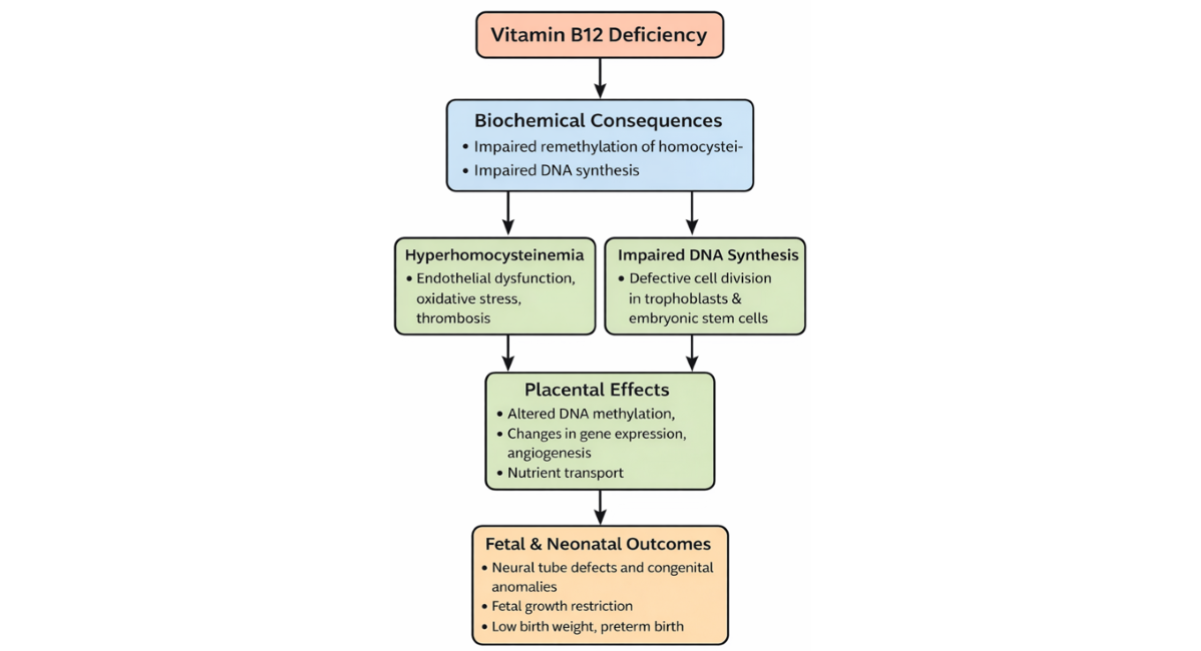
Figure 1: Mechanistic Pathways Linking Maternal Vitamin B12 (Cobalamin) Deficiency to Adverse Pregnancy Outcomes
Legend: Maternal vitamin B12 (cobalamin) deficiency disrupts one-carbon metabolism by impairing the remethylation of homocysteine to methionine and reducing S-adenosylmethionine (SAM) availability. These biochemical alterations lead to hyperhomocysteinemia, endothelial dysfunction, oxidative stress, and defective DNA synthesis in rapidly proliferating trophoblasts and embryonic stem cells. Placental consequences include abnormal DNA methylation, altered gene expression, impaired angiogenesis, and reduced nutrient transport capacity. Collectively, these mechanisms increase the risk of neural tube defects, congenital anomalies, fetal growth restriction, low birth weight, and preterm birth, and may contribute to long-term metabolic programming in offspring.
Maternal B12 Deficiency and MASLD: Emerging Interactions
Metabolic dysfunction-associated steatotic liver disease (MASLD), previously termed non-alcoholic fatty liver disease (NAFLD), is increasingly recognized as an important determinant of adverse pregnancy outcomes, including gestational diabetes mellitus (GDM), preeclampsia, preterm birth, and fetal growth abnormalities [26].
Emerging evidence suggests several potential intersections between maternal B12 deficiency and MASLD:
- Shared metabolic pathways: Vitamin B12 plays a critical role in mitochondrial fatty acid metabolism via its function as a cofactor for methylmalonyl-CoA mutase. Deficiency leads to accumulation of methylmalonic acid and impaired β-oxidation, potentially promoting hepatic lipid accumulation. This metabolic disturbance may exacerbate hepatic steatosis in women predisposed to MASLD.
- Insulin resistance and one-carbon metabolism: Vitamin B12 deficiency disrupts one-carbon metabolism, alters methylation capacity, and may influence genes involved in insulin signaling and lipid regulation. Reduced S-adenosylmethionine (SAM) availability can impair hepatic lipid export and increase oxidative stress, mechanisms central to MASLD progression.
- Hyperhomocysteinemia and endothelial dysfunction: Elevated homocysteine, common in B12 deficiency, contributes to oxidative stress and endothelial dysfunction-key features also implicated in MASLD-related placental insufficiency and adverse pregnancy outcomes.
- Developmental programming: Within the DOHaD framework, maternal MASLD and B12 deficiency may synergistically influence fetal metabolic programming, increasing offspring susceptibility to obesity, insulin resistance, and metabolic syndrome later in life [25].
Although direct interventional evidence remains limited, the overlapping metabolic and epigenetic pathways strongly suggest that optimizing maternal B12 status may represent a modifiable factor in pregnancies complicated by MASLD. Further prospective and mechanistic studies are needed to clarify causality and therapeutic implications.
Diagnosis and Screening Challenges in Pregnancy
Accurate assessment of maternal vitamin B12 status is critical for timely intervention but presents unique diagnostic challenges during pregnancy (Table 2). Serum cobalamin remains the standard initial measurement; however, its sensitivity and specificity are limited in pregnant women. Pregnancy induces physiological changes in B12-binding proteins, particularly transcobalamin, as a result of hormonal fluctuations, often producing a paradoxical decline in total serum B12, even when functional B12 status is adequate [26, 27]. This biological variability underscores the need for more precise functional biomarkers to guide clinical management.
Functional markers that reflect tissue-level B12 status and are less affected by plasma volume changes are increasingly recommended. HoloTC, representing the biologically active fraction of B12 available for cellular uptake, has emerged as a more reliable early indicator of functional deficiency than total serum B12 or methylmalonic acid (MMA) [11]. In a study by Molloy et al. (2009), low HoloTC values predicted poor B12 status more accurately than low total serum B12, with a higher area under the curve (AUC) for HoloTC compared to serum B12 [3]. While MMA is highly specific for tissue deficiency, its interpretation during pregnancy is complicated by altered renal clearance and glomerular filtration rates, which can mask mild deficiency or lower its measured concentration [28].
Additional challenges include the absence of standardized, region-specific cut-offs for these functional markers, particularly in high-prevalence, low-resource settings (LMICs) [29]. This limitation hinders the implementation of effective screening protocols, despite the irreversible nature of fetal neurological damage caused by maternal B12 deficiency. Consequently, there is a strong rationale for universal or targeted screening using HoloTC during the preconception or early antenatal period, particularly in populations with high-risk dietary patterns (e.g., vegetarian or vegan) or in LMICs [3, 29]. Early identification and intervention are essential to mitigate adverse maternal and fetal outcomes and to ensure optimal perinatal and long-term neurodevelopmental health.
Supplementation Strategies and Future Directions
Given the compelling evidence linking maternal vitamin B12 deficiency to adverse reproductive outcomes and impaired fetal neurodevelopment, targeted supplementation is critical [17]. While folic acid supplementation is widely implemented for NTD prevention, current data underscore the urgent need for combined B12 and folate strategies, particularly in regions with high prevalence of B12 deficiency [30].
Efficacy of Supplementation
Randomized controlled trials (RCTs) assessing the clinical impact of maternal B12 supplementation remain relatively limited, yet consistently demonstrate significant biochemical efficacy, especially in high-risk populations [19, 30]. A synthesis of five RCTs involving 984 women in low- and middle-income countries (LMICs) provides quantitative evidence of effective correction of deficiency [11]. Maternal B12 supplementation reduced the prevalence of deficiency from 67.9% to 25.9% (Relative Risk [RR] = 0.38) and increased mean serum concentrations by +60.9 pmol/L. Robust maternal-fetal transfer was confirmed, with infant B12 concentrations increasing by +71.9 pmol/L.
Despite these biochemical successes, evidence for supplementation’s effect on clinical outcomes remains less consistent. For example, pooled effects on preterm birth (RR = 0.97) and low birth weight (RR = 1.50) were minimal or inconclusive. In contrast, the MIND Trial in Nepal, a large RCT of maternal multiple micronutrient supplementation including B12, provided a clear positive signal for offspring neurodevelopment. Children of supplemented mothers demonstrated significant improvements in gross motor and language scores at 2 years compared to controls [32]. These findings highlight that while effects on birth weight remain variable, neurocognitive outcomes are a compelling rationale for targeted maternal B12 intervention.
Current recommendations suggest a daily intake of 2.6 mcg of B12 during pregnancy, though higher doses may be necessary for women with overt deficiency, malabsorption, or strict vegetarian/vegan diets [19].
Conclusion and Clinical Implications
Maternal B12 deficiency, driven by low dietary intake in LMICs and malabsorption in Western populations, represents a significant but often under-recognized determinant of reproductive health. Evidence is strongest for associations with infertility, recurrent pregnancy loss, neural tube defects, and impaired long-term neurodevelopment in offspring [3, 19].
Clinicians should maintain a high index of suspicion, utilizing functional biomarkers such as HoloTC rather than relying solely on total serum B12 for accurate diagnosis [27]. Future research priorities include establishing optimal pregnancy-specific B12 cut-off values, conducting large-scale RCTs to determine the most effective supplementation dose, timing, and delivery strategy, and evaluating long-term offspring outcomes.
Proactive preconception and early antenatal B12 screening represents a cost-effective, preventive intervention with the potential to reduce the global burden of maternal and child morbidity, particularly in high-risk populations. Combined B12 and folate supplementation should be considered a core component of PCT care, addressing both immediate and long-term maternal-fetal health outcomes.
Clinical Recommendations and Future Research Priorities
Women of reproductive age, particularly those belonging to high-risk groups-including vegetarians or vegans, individuals with malabsorption syndromes, or those with a history of pregnancy complications such as recurrent early pregnancy loss-should be proactively screened for vitamin B12 deficiency. Early detection enables timely intervention, which is crucial given the irreversible consequences of prolonged deficiency on fetal neurodevelopment and pregnancy outcomes [3, 33].
Correction of deficiency can be achieved through oral or parenteral B12 supplementation, with consideration of combined B12 and folate administration. This combination leverages their synergistic role in one-carbon metabolism, optimizing DNA synthesis, methylation pathways, and overall reproductive and placental function [19]. Clinicians should adopt functional biomarkers, such as HoloTC, alongside conventional serum B12 measurements to ensure accurate assessment, particularly in populations with physiological variations in B12-binding proteins during pregnancy [27].
From a public health perspective, implementation of targeted supplementation or food fortification programs should be prioritized in high-prevalence regions, such as low-income and middle-income countries or communities with predominantly vegetarian diets, to improve maternal and neonatal outcomes at a population level.
Future research priorities include:
- Well-designed, adequately powered randomized controlled trials to determine optimal B12 dosing, route of administration, and timing for preconception and antenatal supplementation.
- Genetic and mechanistic studies to elucidate causality, identify maternal or fetal genetic polymorphisms affecting B12 metabolism, and clarify complex interactions with folate and homocysteine pathways.
- Development of precision medicine strategies for maternal micronutrient management, integrating biochemical, genetic, and clinical risk factors to individualize supplementation protocols and maximize maternal-fetal health outcomes.
Overall, proactive screening, targeted intervention, and evidence-based supplementation strategies constitute a cost-effective and high-impact approach to reduce the global burden of B12 deficiency-related reproductive and perinatal morbidity.
|
Category |
Gaps in Knowledge |
Clinical Implications |
|
Infertility & Recurrent Pregnancy Loss |
Predominantly observational data; variable definitions of deficiency; limited RCTs |
Screen high-risk women; correct B12 deficiency; consider B12 + folate supplementation |
|
Early Pregnancy Loss (Miscarriage) |
Limited interventional studies; inconsistent biomarkers |
Screening for B12 and homocysteine may help identify at-risk women |
|
Neural Tube Defects & Congenital Anomalies |
Heterogeneous definitions and timing of B12 assessment |
Monitor maternal B12; combined B12 + folic acid supplementation recommended |
|
Preterm Birth & Low Birth Weight |
Lack of long-term follow-up in many studies; population variability |
Early B12 supplementation; public health programs in high-risk populations |
|
Gestational Diabetes Mellitus (GDM) |
Conflicting findings; small sample sizes; variable folate interactions |
Maintain adequate B12; more large-scale, multi-regional studies needed |
|
Overall Research Needs |
Residual confounding, variable biomarkers, inconsistent timing, insufficient long-term offspring data |
Conduct well-powered RCTs and genetic studies to clarify causality, optimal doses, and B12-folate interactions |
|
Public Health |
Limited implementation data for fortification/supplementation programs |
Consider targeted supplementation or fortification programs in high-risk populations |
Table 2: Clinical implications of maternal vitamin B12 and folate status
Experience of the University Hospital of Strasbourg
In a case series at the University Hospital of Strasbourg (France), five women (aged 25-34 years) presenting with infertility and multiple unsuccessful in vitro fertilization (IVF) attempts were evaluated. All were found to have significant B12 deficiency, with a mean B12 level of 140 pmol/L (range 60-140) and elevated homocysteine levels (mean 23 µmol/L, range 17-46). Among them, one patient had anemia with hemoglobin below 10.5 g/dL, and two patients reported paresthesia in the upper extremities. The deficiency was attributed to pernicious anemia in three patients and to food-cobalamin malabsorption in two patients. Following correction of the deficiency through supplementation, these patients underwent subsequent IVF cycles, resulting in successful pregnancies. All pregnancies progressed normally, and the children were born healthy. This clinical observation underscores the potential impact of identifying and correcting maternal B12 deficiency in women with infertility, particularly those undergoing assisted reproductive techniques.
CONCLUSION
A Call for Dual Supplementation in Global Maternal Health
Vitamin B12 deficiency in women of reproductive age represents a neglected but critical determinant of global maternal and child health. Accumulating evidence from epidemiological studies, mechanistic research, and clinical trials demonstrates that insufficient B12 contributes to a spectrum of adverse outcomes, including infertility, recurrent miscarriage, preterm birth, low birth weight, and neural tube defects (NTDs). Mechanistically, B12 deficiency disrupts one-carbon metabolism, leading to hyper Hcyt, impaired DNA synthesis, and altered epigenetic programming, thereby compromising reproductive success and fetal development.
Correction of maternal B12 deficiency is biologically plausible, clinically safe, and potentially transformative. Evidence from supplementation trials demonstrates effective biochemical correction, robust maternal-fetal transfer, and positive signals for offspring neurodevelopment, underscoring the value of timely intervention.
To achieve equitable reproductive health outcomes globally, strategies focusing solely on folate are insufficient. A dual focus on B12 and folate sufficiency should become a cornerstone of preconception and prenatal care, particularly in low-resource settings where nutritional deficiencies are pervasive. Integration of B12 screening-preferably using functional markers such as HoloTC-and targeted supplementation into maternal health policies represents one of the most cost-effective interventions currently available to significantly reduce preventable fetal and neonatal morbidity worldwide.
In summary, global maternal health programs should prioritize combined B12 and folate sufficiency, ensuring that women enter pregnancy with optimal micronutrient status to support fertility, healthy pregnancy outcomes, and long-term child health.
CONFLICT OF INTEREST
The authors declare no conflicts of interest.
ETHICS STATEMENT
According to French legislation (Loi Jarde), retrospective clinical studies based on existing patient records do not require submission to a Comite de Protection des Personnes (CPP) for ethical approval. All patient data used in this case series were derived from paper clinical records maintained in accordance with current French hospital regulations and data-protection standards of Commission Nationale Information Liberte (CNIL). The dataset was fully anonymized, and no identifiable information can be traced back to individual patients; only the clinicians who directly managed the cases had access to the original records. In line with institutional practices at the University Hospitals of Strasbourg, patients are informed that their anonymized clinical data may be used for research and scientific publication purposes. Given these conditions, formal ethics approval was not required for this retrospective, anonymized case series.
ACKNOWLEDGMENT
The authors would like to thank the clinicians and researchers of the CARE B12 (CAREence en B12) group at University Hospital of Strasbourg (France) for their contributions.
The authors gratefully acknowledge the use of various digital tools that facilitated the preparation and finalization of this manuscript. These include ChatGPT (GPT-5 Mini, OpenAI) for text drafting and refinement, Gemini for research assistance, Zotero for reference management, PubMed for literature retrieval, and the Microsoft Office Suite for document processing and formatting.
Reason for Using AI: AI tools were employed to enhance the clarity, coherence, and scientific precision of the manuscript text, assist in structuring complex sections, and generate illustrative content concepts for figures. Specific AI Used: ChatGPT (GPT-5 Mini, OpenAI, Gemini) - for text, coherence text drafting, language refinement (the authors were not native English speakers), and figure concept development.
REFERENCES
- Andrès E, Vogel T, Federici L, Zimmer J, Ciobanu E, Kaltenbach G. Cobalamin deficiency in elderly patients: a personal view. Curr Gerontol Geriatr Res. 2008;2008:848267. [Google Scholar] [PubMed] [Crossref]
- Wang L, Hou Y, Meng D, Yang L, Meng X, Liu F. Vitamin B12 and Folate Levels During Pregnancy and Risk of Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Front Nutr. 2021;8:670289. [Google Scholar] [PubMed] [Crossref]
- Molloy AM, Kirke PN, Brody LC, Scott JM, Mills JL. Effects of folate and vitamin B12 deficiencies during pregnancy on fetal, infant, and child development. Food Nutr Bull. 2008;29(2 Suppl):S101-S111. [Google Scholar] [PubMed] [Crossref]
- The Lancet Global Health. Global health 2021: who tells the story?. Lancet Glob Health. 2021;9(2):e99. [Google Scholar] [Crossref]
- Bjørkevoll SM, Konijnenberg C, Kvestad I, McCann A, Ueland PM, Næss Sleire S, et al. Vitamin B12 and Folate Status in Pregnant Females and Their Infants in Norway: Secondary Analysis from the Mommy's Food Study. J Nutr. 2023;153(12):3543-3554. [Google Scholar] [PubMed] [Crossref]
- O'Malley EG, Reynolds CME, Cawley S, Woodside JV, Molloy AM, Turner MJ. Folate and vitamin B12 levels in early pregnancy and maternal obesity. Eur J Obstet Gynecol Reprod Biol. 2018;231:80-84. [Google Scholar] [PubMed] [Crossref]
- Zhang L, Wu LM, Xu WH, Tian YQ, Liu XL, Xia CY, Zhang L, Li SS, Jin Z, Wu XL, Shu J. Status of maternal serum B vitamins and pregnancy outcomes: New insights from in vitro fertilization and embryo transfer (IVF-ET) treatment. Front Nutr. 2022;9:962212. [Google Scholar] [PubMed] [Crossref]
- Revelli A, Delle Piane L, Casano S, Molinari E, Massobrio M, Rinaudo P. Follicular fluid content and oocyte quality: from single biochemical markers to metabolomics. Reprod Biol Endocrinol. 2009;7:40. [Google Scholar] [PubMed] [Crossref]
- Gaskins AJ, Chiu YH, Williams PL, Ford JB, Toth TL, Hauser R, Chavarro JE; EARTH Study Team. Association between serum folate and vitamin B-12 and outcomes of assisted reproductive technologies. Am J Clin Nutr. 2015;102(4):943-950. [Google Scholar] [PubMed] [Crossref]
- Bala R, Verma R, Verma P, Singh V, Yadav N, Rajender S, Agrawal NR, Singh K. Hyperhomocysteinemia and low vitamin B12 are associated with the risk of early pregnancy loss: A clinical study and meta-analyses. Nutr Res. 2021;91:57-66. [Google Scholar] [PubMed] [Crossref]
- Dai C, Fei Y, Li J, Shi Y, Yang X. A Novel Review of Homocysteine and Pregnancy Complications. Biomed Res Int. 2021;2021:6652231. [Google Scholar] [PubMed] [Crossref]
- Nie L, Liu X, Li X, Ren Z, Cheng X, Wu Y, Li Z, Liu J. Beyond Folate: The Emerging Role of Maternal Vitamin B12 in Neural Tube Development. Nutrients. 2025;17(12):2040. [Google Scholar] [PubMed] [Crossref]
- Wang X, Fu K, Zhang Y, Jiang S, Zhang C. Does vitamin B12 influence clinical pregnancy outcome in the follicular-phase GnRH agonist protocol?. BMC Pregnancy Childbirth. 2025;25(1):1120. [Google Scholar] [PubMed] [Crossref]
- Rogne T, Tielemans MJ, Chong MFF, Yajnik CS, Krishnaveni GV, Poston L, et al. Associations of Maternal Vitamin B12 Concentration in Pregnancy With the Risks of Preterm Birth and Low Birth Weight: A Systematic Review and Meta-Analysis of Individual Participant Data. Am J Epidemiol. 2017;185(3):212–223. [Google Scholar] [PubMed] [Crossref]
- Sukumar N, Rafnsson SB, Kandala NB, Bhopal R, Yajnik CS, Saravanan P. Prevalence of vitamin B-12 insufficiency during pregnancy and its effect on offspring birth weight: a systematic review and meta-analysis. Am J Clin Nutr. 2016;103(5):1232-1251. [Google Scholar] [PubMed] [Crossref]
- Finkelstein JL, Kurpad AV, Thomas T, Srinivasan K, Duggan C. Vitamin B12 status in pregnant women and their infants in South India. Eur J Clin Nutr. 2017;71(9):1046-1053. [Google Scholar] [PubMed] [Crossref]
- Behere RV, Deshmukh AS, Otiv S, Gupte MD, Yajnik CS. Maternal Vitamin B12 Status During Pregnancy and Its Association With Outcomes of Pregnancy and Health of the Offspring: A Systematic Review and Implications for Policy in India. Front Endocrinol. 2021;12:619176. [Google Scholar] [PubMed] [Crossref]
- Srinivasan K, Thomas T, Kapanee AR, Ramthal A, Bellinger DC, Bosch RJ, Kurpad AV, Duggan C. Effects of maternal vitamin B12 supplementation on early infant neurocognitive outcomes: a randomized controlled clinical trial. Matern Child Nutr. 2017;13(2):e12325. [Google Scholar] [PubMed] [Crossref]
- Finkelstein JL, Fothergill A, Venkatramanan S, Layden AJ, Williams JL, Crider KS, Qi YP. Vitamin B12 supplementation during pregnancy for maternal and child health outcomes. Cochrane Database Syst Rev. 2024(1). [Google Scholar] [PubMed] [Crossref]
- Wang L, Hou Y, Meng D, Yang L, Meng X, Liu F. Vitamin B12 and Folate Levels During Pregnancy and Risk of Gestational Diabetes Mellitus: A Systematic Review and Meta-Analysis. Front Nutr. 2021;8:670289. [Google Scholar] [PubMed] [Crossref]
- Arcot A, Walker RE, Gallagher K, Klatt KC, Gernand AD. Maternal Vitamin B12 in Pregnancy and Placental Development. J Nutr Metab. 2024;2024:3439995. [Google Scholar] [PubMed] [Crossref]
- Memon SI, Acharya NS. The Association Between Serum Homocysteine Levels and Placenta-Mediated Complications: A Narrative Review. Cureus. 2022;14(11):e31305. [Google Scholar] [PubMed] [Crossref]
- Sapehia D, Mahajan A, Srinivasan R, Kaur J. Pre-natal dietary imbalance of folic acid and vitamin B12 deficiency adversely impacts placental development and fetal growth. Placenta. 2023;132:44-54. [Google Scholar] [PubMed] [Crossref]
- van Vliet MM, Schoenmakers S, Willemsen SP, Sinclair KD, Steegers-Theunissen RPM. First-trimester maternal folate and vitamin B12 concentrations and their associations with first-trimester placental growth: the Rotterdam Periconception Cohort. Hum Reprod. 2025;40(8):1485-1494. [Google Scholar] [PubMed] [Crossref]
- Lacagnina S. The Developmental Origins of Health and Disease (DOHaD). Am J Lifestyle Med. 2019;14(1):47-50. [Google Scholar] [PubMed] [Crossref]
- Yang M, Xiao X, Mei J, Gong Q. MASLD: insights on the role of folate in hepatic lipid metabolism. Front. Nutr. 2025;12:1583674. [Google Scholar] [PubMed] [Crossref]
- Greibe E, Andreasen BH, Lildballe DL, Morkbak AL, Hvas AM, Nexo E. Uptake of cobalamin and markers of cobalamin status: a longitudinal study of healthy pregnant women. Clin Chem Lab Med. 2011;49(11):1877-1882. [Google Scholar] [PubMed] [Crossref]
- Dib MJ, Gumban-Marasigan M, Yoxall R, Andrew T, Harrington DJ, Sobczyńska-Malefora A, Ahmadi KR. Evaluating the Diagnostic Value of a Combined Indicator of Vitamin B12 Status (cB12) Throughout Pregnancy. Front Nutr. 2022;8:789357. [Google Scholar] [PubMed]
- Lehto E, Kaartinen NE, Sääksjärvi K, Männistö S, Jallinoja P. Vegetarians and different types of meat eaters among the Finnish adult population from 2007 to 2017. British Journal of Nutrition. 2022;127(7):1060-1072. [Google Scholar] [PubMed] [Crossref]
- Pentieva K, Caffrey A, Duffy B, et al. B-vitamins and one-carbon metabolism during pregnancy: health impacts and challenges. Proc Nutr Soc. 2025;84(3):264-278. [Google Scholar] [PubMed] [Crossref]
- Duggan C, Srinivasan K, Thomas T, Samuel T, Rajendran R, Muthayya S, Finkelstein JL, Lukose A, Fawzi W, Allen LH, Bosch RJ, Kurpad AV. Vitamin B-12 supplementation during pregnancy and early lactation increases maternal, breast milk, and infant measures of vitamin B-12 status. J Nutr. 2014;144(5):758-764. [Google Scholar] [PubMed] [Crossref]
- Chandyo RK, Kvestad I, Ulak M, Ranjitkar S, Hysing M, Shrestha M, Schwinger C, McCann A, Ueland PM, Basnet S, Shrestha L, Strand TA . The effect of vitamin B12 supplementation during pregnancy on infant growth and development in Nepal: a community-based, double-blind, randomised, placebo-controlled trial. Lancet. 2023 May 6;401(10387):1508-1517. [Google Scholar] [PubMed] [Crossref]
- Sebastiani G, Herranz Barbero A, Borrás-Novell C, Alsina Casanova M, Aldecoa-Bilbao V, et al. The Effects of Vegetarian and Vegan Diet during Pregnancy on the Health of Mothers and Offspring. Nutrients. 2019;11(3):557. [Google Scholar] [PubMed] [Crossref]