ABSTRACT
Cassava mosaic disease (CMD) is among key threats to cassava (Manihot esculenta Crantz) production in Sierra Leone. A diagnostic survey was conducted in 2022 to determine the status of cassava mosaic begomoviruses, whitefly abundance and alternate hosts in cassava field surveys of Sierra Leone. A total of 278 cassava farms were surveyed following the harmonized protocol of the Central and West African Virus Epidemiology program. Findings revealed that, of the 278 farms visited, 10 (3.5%) had no visible symptom of the disease. Of the 8,281 cassava leaf samples assessed, 7981 had symptoms. Average CMD incidence was 90.5%, with the Western Area region recording the highest severity score (3.7), followed by the Northern region (3.2), and the Southern region had the lowest of 2.4. Cutting-borne infection was identified as the primary source of CMD infection. The average whitefly count per field was 5.17. The relationship between CMD incidence and whitefly abundance was significantly negative, while the relationship between CMD severity and incidence was significantly positive. Most farms had ACMV in single and in mixed infections with EACMV and EACMCMV. Most doubly infected plants showed severe symptoms. The ACMV and EACMV were also detected in putative natural hosts Centrosema pubescens, Senna occidentalis, Carica papaya, and Manihot glaziovii of the viruses. The occurrence of variants of ACMV and a high proportion of mixed infections by ACMV, EACMV and EACMCMV, could result in recombination events, suggesting appropriate measures to safe guard cassava production in Sierra Leone.
Keywords: Begomoviruses Status; Distribution and Spread; Damage; whitefly Abundance; Natural hosts; Manihot esculenta
INTRODUCTION
Cassava (Manihot esculenta Crantz) is a highly significant root crop, widely utilized for human consumption, animal feed, and industrial applications [1]. The starchy storage roots of cassava have emerged as a crucial source of dietary energy in sub-Saharan Africa (SSA), offering higher returns per unit of input compared to other staple crops [1, 2]. Cassava leaves, containing high protein levels (17-18%), are also useful as a food source in Africa [3]. Cassava exhibits exceptional resilience, thriving in nutrient-poor soils and displaying superior yield performance compared to other root and tuber crops. In Sierra Leone, cassava is the second most important food crop after rice.
Despite its crucial role in providing food and income in many developing countries including Sierra Leone, cassava production faces challenges from biotic factors like Cassava Mosaic Disease (CMD) and insect pests [4]. The response of cassava genotypes to diverse environmental factors (such as soil and climate) and biotic stresses vary significantly [5]. The country's average storage root cassava yield is low, estimated at 7.18 metric tonnes per hectare compared to Africa's average of 8.4 metric tonnes per hectare [6]. The low storage root yield of cassava is attributable to several production-limiting factors, including diseases, pests, weed infestation, and edaphic, agronomic, and socio-economic factors [7]. Diseases play a prominent role in limiting cassava productivity in Sierra Leone, with economically important ones including CMD, cassava bacterial blight (CBB), and recently cassava brown streak virus.
The CMD is endemic to Africa and is caused by at least seven different species of whitefly (Bemisia tabaci) transmitted geminiviruses, commonly referred to as cassava mosaic geminiviruses (CMGVs) [8]. The CMD caused by several viruses that belong to the genus Begomovirus of the family Geminiviridae [9]. These viruses are likely descendants of geminiviruses adapted to infect indigenous uncultivated African plant species [10]. Eight out of 17 of these viruses that affect cassava are endemic to Africa [9]. CMD is a major constraint to cassava production in Africa [11]. Scientists in Africa have drawn attention to the types of viruses causing cassava mosaic disease and cassava brown streak diseases, emphasizing the inadequate attention given to the significant losses caused by these viruses to African farmers [12].
Several species and strains of cassava mosaic geminivirus have been described. However, limited information on cassava mosaic viruses has been provided from Sierra Leone in terms of their distribution and effects on growth or yield [13]. These are serious deficiencies, emphasizing the inadequate attention given to cassava mosaic viruses in Sierra Leone, which affect the most important storage root crop. The traditional belief that mosaic-infected cassava genotypes act as indicators for identifying poundable cassava genotypes with high dry matter content has encouraged the use of locally infected genotypes. Furthermore, there is a popular demand for mildly mosaic-infected cassava leaves among women for preparing a popular vegetable sauce [7]. It is believed that the infected leaves, due to their low chlorophyll content, consume less palm oil an expensive but crucial element in the preparation of cassava leaf sauce compared to improved resistant varieties, which consume more. Women play a vital role in selecting planting materials and often favor infected local varieties for domestic consumption. Preliminary surveys indicated that ACMV is prevalent throughout the western areas of Sierra Leone with an estimated 100% incidence in smallholder farms. Spot checks on farmers’ fields in other parts of the country, such as Bo, Kemena, Pujehun, and Njala, indicate similar trends, warranting a nationwide survey on the prevalence and distribution of the mosaic virus as well as the diversity of the CMGVs [14]. The large molecular diversity of viruses from East Africa points to the region as the center of diversification. Additionally, recombination events evident in many virus genomes drive geminivirus evolution [15]. Virus diversity and frequent recombination events provide evidence for continuous evolutionary processes, influencing the development of epidemics and the emergence of new viruses [15]. Knowledge of virus diversity, geographic distribution of virus types, and virus population structure is a significant prerequisite to deploy cassava with virus-resistant characteristics. Various species and strains of cassava mosaic geminivirus have been identified [15].
Previous survey on CMD revealed that farms with local cassava varieties were more numerous than those with improved varieties. The study found that the prevalence of CMD across the country was 85.2% among 156 sites visited, with mixed infections of African Cassava Mosaic Virus and East African Cassava Mosaic Virus present in infected cassava genotypes [14]. Financial losses due to disease are huge and rising across the continent; estimates made some two decades ago put the loss at billions of dollars [16]. Many local cultivars are already infected with CMD [17, 18] and the disease is widespread across the country necessitating the present study. Thus, the objective of this study was to determine the status of cassava mosaic disease, whitefly population dynamics and alternate hosts in cassava growing regions of Sierra Leone.Cassava (Manihot esculenta Crantz) is a highly significant root crop, widely utilized for human consumption, animal feed, and industrial applications [1]. The starchy storage roots of cassava have emerged as a crucial source of dietary energy in sub-Saharan Africa (SSA), offering higher returns per unit of input compared to other staple crops [1, 2]. Cassava leaves, containing high protein levels (17-18%), are also useful as a food source in Africa [3]. Cassava exhibits exceptional resilience, thriving in nutrient-poor soils and displaying superior yield performance compared to other root and tuber crops. In Sierra Leone, cassava is the second most important food crop after rice.
Despite its crucial role in providing food and income in many developing countries including Sierra Leone, cassava production faces challenges from biotic factors like Cassava Mosaic Disease (CMD) and insect pests [4]. The response of cassava genotypes to diverse environmental factors (such as soil and climate) and biotic stresses vary significantly [5]. The country's average storage root cassava yield is low, estimated at 7.18 metric tonnes per hectare compared to Africa's average of 8.4 metric tonnes per hectare [6]. The low storage root yield of cassava is attributable to several production-limiting factors, including diseases, pests, weed infestation, and edaphic, agronomic, and socio-economic factors [7]. Diseases play a prominent role in limiting cassava productivity in Sierra Leone, with economically important ones including CMD, cassava bacterial blight (CBB), and recently cassava brown streak virus.
The CMD is endemic to Africa and is caused by at least seven different species of whitefly (Bemisia tabaci) transmitted geminiviruses, commonly referred to as cassava mosaic geminiviruses (CMGVs) [8]. The CMD caused by several viruses that belong to the genus Begomovirus of the family Geminiviridae [9]. These viruses are likely descendants of geminiviruses adapted to infect indigenous uncultivated African plant species [10]. Eight out of 17 of these viruses that affect cassava are endemic to Africa [9]. CMD is a major constraint to cassava production in Africa [11]. Scientists in Africa have drawn attention to the types of viruses causing cassava mosaic disease and cassava brown streak diseases, emphasizing the inadequate attention given to the significant losses caused by these viruses to African farmers [12].
Several species and strains of cassava mosaic geminivirus have been described. However, limited information on cassava mosaic viruses has been provided from Sierra Leone in terms of their distribution and effects on growth or yield [13]. These are serious deficiencies, emphasizing the inadequate attention given to cassava mosaic viruses in Sierra Leone, which affect the most important storage root crop. The traditional belief that mosaic-infected cassava genotypes act as indicators for identifying poundable cassava genotypes with high dry matter content has encouraged the use of locally infected genotypes. Furthermore, there is a popular demand for mildly mosaic-infected cassava leaves among women for preparing a popular vegetable sauce [7]. It is believed that the infected leaves, due to their low chlorophyll content, consume less palm oil an expensive but crucial element in the preparation of cassava leaf sauce compared to improved resistant varieties, which consume more. Women play a vital role in selecting planting materials and often favor infected local varieties for domestic consumption. Preliminary surveys indicated that ACMV is prevalent throughout the western areas of Sierra Leone with an estimated 100% incidence in smallholder farms. Spot checks on farmers’ fields in other parts of the country, such as Bo, Kemena, Pujehun, and Njala, indicate similar trends, warranting a nationwide survey on the prevalence and distribution of the mosaic virus as well as the diversity of the CMGVs [14]. The large molecular diversity of viruses from East Africa points to the region as the center of diversification. Additionally, recombination events evident in many virus genomes drive geminivirus evolution [15]. Virus diversity and frequent recombination events provide evidence for continuous evolutionary processes, influencing the development of epidemics and the emergence of new viruses [15]. Knowledge of virus diversity, geographic distribution of virus types, and virus population structure is a significant prerequisite to deploy cassava with virus-resistant characteristics. Various species and strains of cassava mosaic geminivirus have been identified [15].
Previous survey on CMD revealed that farms with local cassava varieties were more numerous than those with improved varieties. The study found that the prevalence of CMD across the country was 85.2% among 156 sites visited, with mixed infections of African Cassava Mosaic Virus and East African Cassava Mosaic Virus present in infected cassava genotypes [14]. Financial losses due to disease are huge and rising across the continent; estimates made some two decades ago put the loss at billions of dollars [16]. Many local cultivars are already infected with CMD [17, 18] and the disease is widespread across the country necessitating the present study. Thus, the objective of this study was to determine the status of cassava mosaic disease, whitefly population dynamics and alternate hosts in cassava growing regions of Sierra Leone.
2. Materials and Methods
2.1 Description of the study area
The study was carried out in Sierra Leone of which there were two surveys and two experiments conducted. The first survey was conducted across Sierra Leone (Figure 1). Sierra Leone, a country located on the west coast of Africa between latitudes 7° and 10° N and longitudes 10° and 13° W, offers a rich and complex field of study encompassing disciplines such as history, political science, anthropology, economics, environmental studies, and public health. Covering a total land area of approximately 71,740 km2 (27,699 square miles), Sierra Leone boasts a diverse landscape with a wide array of geographical features important for survey studies. The western region features flat coastal plains extending inland for about 100 km, characterized by mangrove swamps and river deltas. As one moves inland, the terrain becomes more rugged, with the central and eastern parts of the country dominated by hills and mountains. The Loma Mountains, including the highest peak, Mount Bintumani at 1,945 m, provide significant elevation. Dense tropical rainforests cover much of the eastern region, contributing to biodiversity and ecological studies.
Sierra Leone is bordered by Guinea to the north and northeast, with a border length of approximately 652 km, and by Liberia to the southeast, with a border length of about 306 km. The Atlantic Ocean forms the western boundary, providing a coastline of about 402 km, which is critical for marine and coastal surveys. The country experiences a tropical climate with a wet season from May to November characterized by heavy rains, receiving annual rainfall ranging from 2,000 to 3,000 mm, with the coastal areas receiving the highest amounts. The dry season, from December to April, is marked by lower precipitation and the harmattan winds, which can affect visibility and survey conditions.
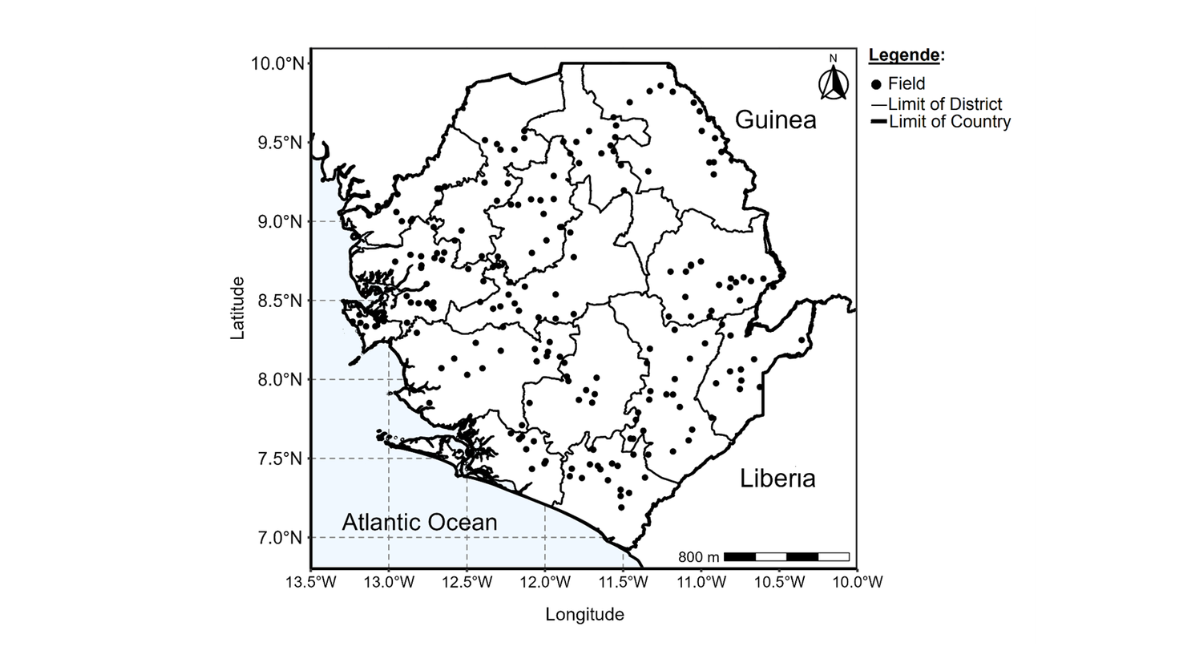
Figure 1: Locations of the 278 farms surveyed in the Northern, Eastern Southern and Western Area for CMD in 2022/2023 cropping season
2.2 Survey design
Cassava mosaic disease assessment survey was conducted in 2022/2023 cropping season using the Central and West African Virus Epidemiology (WAVE) harmonized sampling and diagnostic protocols [19, 20]. During the survey, data and cassava leaf, whitefly vector samples were collected from plants with age between 3-6 months. Assessments were done in 268 farms/fields surveyed in all five regions and sixteen districts which covers all the agro ecologies (rain forest, coastal plains, savannah lowlands and savannah highlands) of Sierra Leone. Cassava fields within villages and towns were sampled with distance between survey sites varied generally 10-20 km or availability of cassava farms.
2.3 Data recording and storage
Tablet with iForm Zerion (version 9.1.6) software developed by Cambridge, UK’s Epidemiology modelling group for survey in all West Africa virus epidemiology countries was used to record data in each survey site. Data collected included name of the village or town, the district, region, whitefly counts, cassava mosaic disease symptom observed, geographical coordinates (latitude and longitude), mode of infection and attitude. Other data on variety, date and time, field size, planting types and distance between survey site was collected. The recorded data were uploaded to iForm’s cloud-based database and then integrated into the WAVE Cube. A total of 30 cassava plants were assessed along two diagonals in an X shape (15 plants chosen randomly on each diagonal). On each selected plant, data were collected on CMD severity, whitefly abundance and where the plant was infected, the source of infection was determined as either from cuttings or by the vector. According to Sseruwagi et al. [21] from three to six MAP, a distinction is possible between cutting-borne and whitefly-borne infections. Symptoms appearing only on upper leaves were taken to have resulted from whitefly-transmitted infection, whereas plants that showed symptoms either only on the lower leaves or on all leaves were taken as having been infected through cassava cuttings.
Leaf samples of weeds showing characteristic symptoms of mosaic in or near the cassava fields also were collected. During the survey trip, leaf samples were kept at 4ºC in a mobile refrigerator (model V 200; Norcold Sidney, a subsidiary of Thetford Corporation, Ann Arbor, MI). The samples were stored at –20ºC in the laboratory during analysis.
2.4 Disease Assessment
The severity of the symptoms was recorded using the standard scale of 1 = symptom-less plants; severity score 2 = mild chlorotic patterns affecting most leaves, mild distortions at the bases of most leaves and remaining part of the leaves are normal; severity score 3 = pronounced chlorosis on most leaves, narrowing and distortion of the lower one-third of the leaflets; severity score 4 = severe chlorosis and distortion of two-thirds of most leaves and general reduction of leaf size and some stunting; and severity score 5 = most severe symptoms (severe chlorosis, leaf distortion, twisting, misshapen leaves, severe reduction of most leaves and severe plant stunting) [22] (Figure 2). The CMD incidence was calculated as the percentage of CMD symptomatic plants out of the total plants assessed using the formula provided by Sseruwagi et al. [21] below.
The incidence was then visually categorized into five percentage: fields with 0 incidence were recorded as healthy; >0–25% as low incidence; >25–50% as medium incidence; >50–75% as high incidence; and >75–100% as very high incidence.
The whitefly population was estimated by counting the number of whiteflies on the top five fully expanded leaves of each plant. The mean of whiteflies per plant was calculated as the total number of whiteflies recorded on 30 plants divided by 30. The mode of infection in each plant was determined based on the location of the symptomatic leaves as previously described by Sseruwagi et al. [21].

Figure 2: Cassava mosaic disease symptoms in survey fields: a=asymptomatic plants; b=mild infection; c=moderate infection; d=severe infection; and d=very severe infection
DNA sample collection and extraction. A total of 1,065 samples comprising 1,055 cassava leaf samples and 10 alternate host samples were collected during the survey. The samples were labelled and preserved following the WAVE harmonized protocol. Total DNA was extracted from the cassava and weed samples using the CTAB protocol [23]. DNA concentration was carried out using a NanoDrop TM 2000 spectrophotometer (Thermo Fisher Scientific Inc., Waltham, MA 02451, Massachusetts, USA) by adjusting the equipment to 150 ng/μL. Previous studies noted that the most cumbersome CMBs in smallholder cassava cultivation systems in Sierra Leone were ACMV and East African cassava mosaic virus (EACMV) [23]. For detection of the ACMV-like and EACMV-like viruses, the DNA samples were subjected to PCR using specific primers (Table 3.2.1). The positive samples for the EACMV-like virus were subjected to another round of PCR using specific primers to detect EACMVCM. The PCR mix was prepared in a final volume of 25 μL using 20.9 μL of molecular biology grade water, 2.5 µL of 10× reaction buffer, 0.5 µL of 10 mM dNTPs, 0.5 µL of 10 µM of each primer, 0.1 µL of 5 U/µL of Maximo Taq DNA polymerase (GeneON), and 150 ng DNA template of each sample. DNA amplification was carried out in a SimpliAmp thermal cycler (Life Technologies Holdings Pte Ltd., Marsiling Industrial Estate, Marsiling, Singapore). The PCR temperature profile was set at 94 °C for 4 or 5 min for initial denaturation, followed by 35 cycles of amplification at 94 °C for 45 or 60 s, 55 °C for 45 or 60 s, and 72 °C for 55 or 60 s (depending on primers). The final elongation step was performed at 72 °C for 7 or 10 min. The PCR-amplified products were subjected to 1% agarose gel electrophoresis and then stained with ethidium bromide. The electrophoresis was performed at 100 V and the gel was visualized using a Compact Digimage System, UVDI series (MS major science). The DNA extraction buffer and DNA of virus-free healthy cassava were used as negative controls.
Table 1: Details of primer pairs utilized for detection of virus species
|
Primer |
Sequence (5′-3’) |
Target region |
Expected size (bp) |
Virus species |
Reference |
|
JSP001 |
ATGTCGAAGCGACCAGGAGAT |
DNA-A (CP) |
783 |
ACMV |
Pita et al. [21] |
|
JSP002 |
TGTTTATTAATTGCCAATACT |
||||
|
ACMBVF |
TCGGGAGTGATACATGCGAAGGC |
DNA-B (BV1/BC1) |
628 |
ACMV |
Matic et al. [25] |
|
ACMBVR |
GGCTACACCAGCTACCTGAAGCT |
||||
|
WAVE-508F |
AAGGCCCATGTAAGGTCCAG |
AV1/AC3 |
800 |
ACMV |
WAVE |
|
WAVE-1307R |
GAAGGAGCTGGGGATTCACA |
||||
|
WAVE-177F |
GATCTGCGGGCCTATCGAAT |
BV1 |
800 |
ACMV |
WAVE |
|
WAVE-197R |
TTCACGCTGTGCAATACCCT |
||||
|
WAVE-370F |
ACAGCCCATACAGGAACCGT |
AV1/AC3 |
1000 |
ACMV |
WAVE |
|
WAVE-1369R |
CGACCATTCCTGCTGAACCA |
||||
|
WAVE-982F |
TTCGTGTCATCTGCAGGAGA |
BV1/BC1 |
800 |
ACMV |
WAVE |
|
WAVE-1781R |
GTACCATGGCAGCTGCTGTA |
||||
|
JSP001 |
ATGTCGAAGCGACCAGGAGAT |
DNA-A (CP) |
780 |
EACMV |
Pita et al. [24] |
|
JSP003 |
CCTTTATTAATTTGTCACTGC |
||||
|
CMBRepF |
CRTCAATGACGTTGTACCA |
DNA-A (AC1) |
650 |
EACMV |
Alabi et al. [26] |
|
EACMVRepR |
GGTTTGCAGAGAACTACATC |
||||
|
WAVE-EA1875F |
TGTACCAGGCGTCGTTTGAA |
AC1 |
800 |
EACMVCM |
WAVE |
|
WAVE-E2674R |
TGTCCCCCGATCCAAAACG |
||||
|
WAVE-EB1869F |
TTCCAAGGGGAGGGTTCTGA |
BC1 |
800 |
EACMV |
WAVE |
For weed samples ACMBVF/ACMBVR and WAVE-508F/WAVE-1307R were used
3.0 Results
3.1 Status of CMD in Survey fields across Sierra Leone
The survey assessed 8,487 plants, of which, 2,462 plants had mild infection of CDM, 4,420 plants had low severity, 1,145 plants were moderate and 160 plants had high infection of the disease (Table 2). A greater number of the plants assessed 8,152 (98.4%) were infected through contaminated cuttings and only 129 (1.6%) of the plants assessed were found to be infected by whitefly.
Table 2: Cassava mosaic disease severity, number of plants assessed and mode of infection
|
CMD severity level |
Number of plants assessed |
Cutting derived infection |
Whitefly-derived infection |
|
Severity 1 |
300 |
- |
- |
|
Severity 2 |
2,462 |
2,438 |
24 |
|
Severity 3 |
4,420 |
4,409 |
11 |
|
Severity 4 |
1,145 |
1,145 |
94 |
|
Severity 5 |
160 |
160 |
0 |
The assessment further revealed that 167 (60.2%) out of 278 fields record very high CMD incidence (above 75% to 100%), 57 fields (20.5%) recorded high CMD incidence (50-75%), 32 (11.5%) fields recorded medium CMD incidence (> 25-50%), 12 fields (4.3%) recorded low CMD incidence (0-25%) (Figure 3). Out of the 278 fields assessed, only 10 fields were healthy (3.5) and they were mostly found in Bonthe, Falaba and Bombali districts. This number was however higher than what was recorded in 2020 survey.
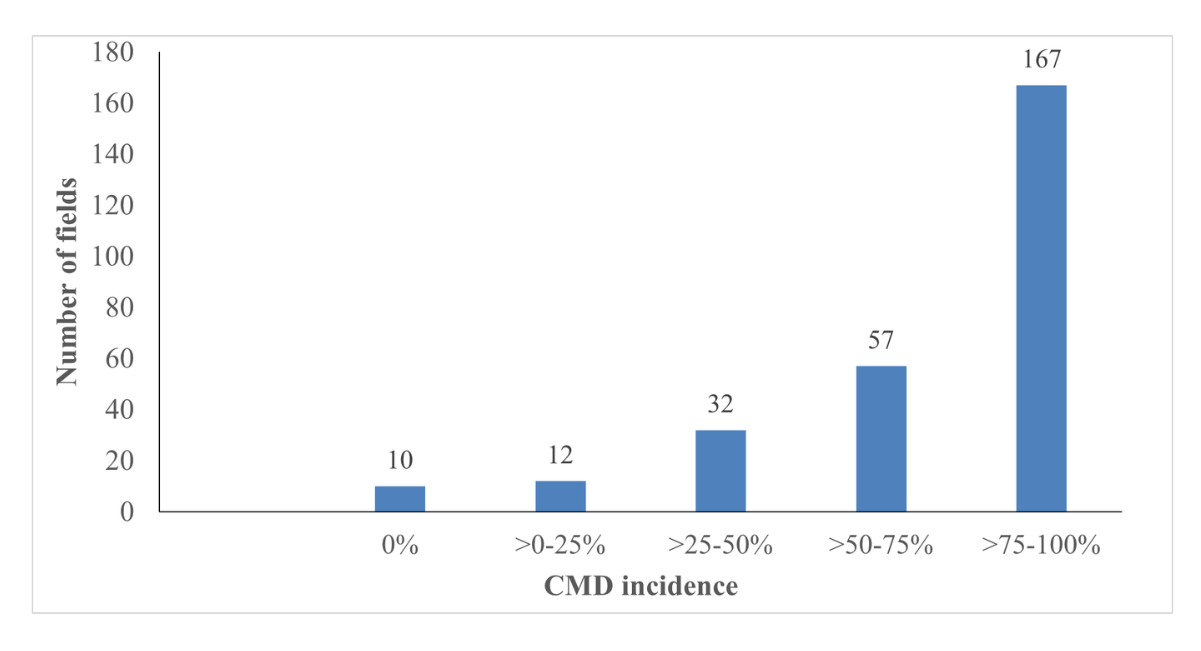
Figure 3: Number of fields by cassava mosaic disease incidence
Cassava mosaic disease incidence and severity per region and district in Sierra Leone
The CMD incidence was very high in many surveyed fields (240) and across the country with a mean CMD incidence of 90.5%. At regional level, the CMD incidence ranges from 66.6% in the Southern region to 99.8% in the Western area (Table 3). The lowest CMD incidence (22.0%) was observed in Moyamba district and the highest CMD incidence (100.0%) was observed in Kono and Western area urban district at district level evaluation. The highest CMD incidence observed in Kono district in the Eastern region and Western Area urban district in the Western Area region were consistent with the CMD incidence in the other district of the same regions. Western area region even recorded the highest CMD incidence at regional level evaluation. The findings also observed widespread distribution of CMD incidence across the country as indicated in the distribution map (Figure 4).
Table 3: Cassava mosaic disease incidence and severity per region and district in Sierra Leone
|
Region |
District |
CMD incidence |
CMD Severity |
|
Eastern |
99.4±6.5 |
3.1±0.3 |
|
|
Kenema |
98.5±4.5 |
3.1±0.2 |
|
|
Kailahun |
99.9±4.5 |
3.3±0.3 |
|
|
Kono |
100.0±6.8 |
3.0±0.3 |
|
|
Southern |
66.6±1.9 |
2.4±0.2 |
|
|
Bo |
74.5±4.6 |
2.4±0.2 |
|
|
Bonthe |
71.4±2.8 |
3.0±0.0 |
|
|
Moyamba |
22.0±1.3 |
2.8±0.1 |
|
|
Pujehun |
98.8±4.5 |
3.5±0.3 |
|
|
Northern |
94.1±5.2 |
3.2±0.1 |
|
|
Bombali |
84.5±5.2 |
2.8±0.0 |
|
|
Tonkolili |
93.5±7.0 |
3.1±0.2 |
|
|
Koinadugu |
98.8±4.5 |
3.5±0.3 |
|
|
Falaba |
99.7±4.3 |
4.1±2.0 |
|
|
Northwest |
92.9±8.2 |
2.9±0.0 |
|
|
Karena |
98.8±4.5 |
3.5±0.2 |
|
|
Portloko |
84.5±2.2 |
3.0±0.2 |
|
|
Kambia |
95.5±4.0 |
3.3±0.3 |
|
|
Western Area |
99.8±4.3 |
3.7±0.3 |
|
|
Western rural |
99.7±7.7 |
3.3±0.0 |
|
|
Western urban |
100.0±9.0 |
3.1±0.3 |
|
|
Mean±SE |
90.5±7.3 |
3.0±0.2 |
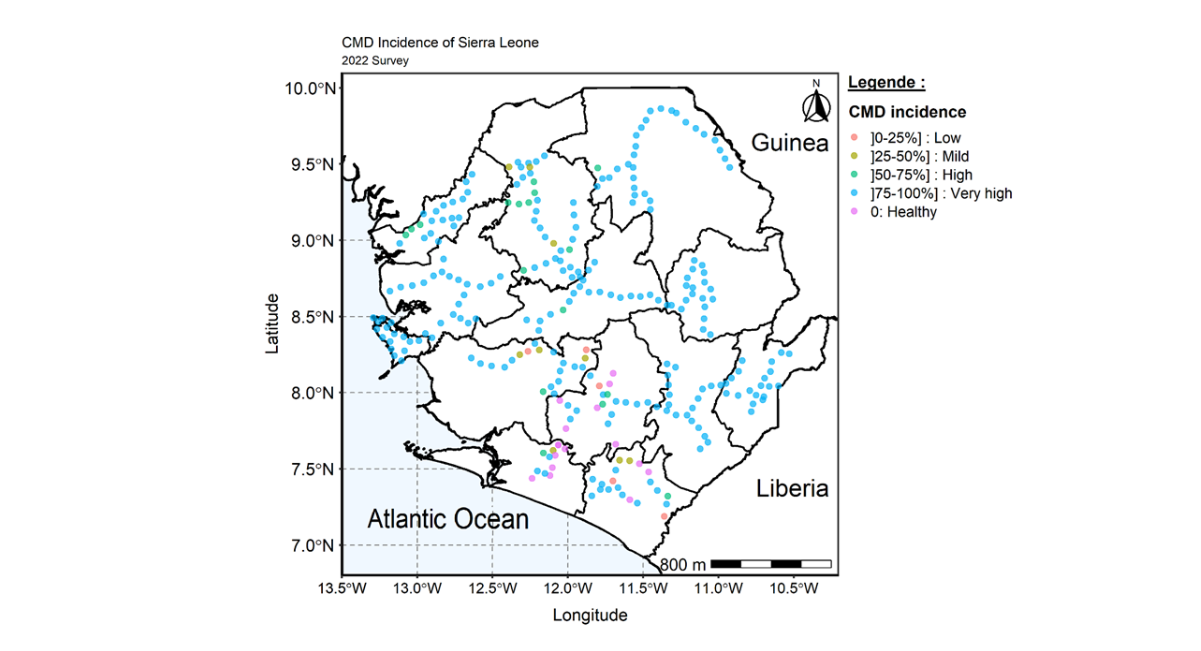
Figure 4: Map of Sierra Leone showing the distribution and spread of CMD incidence across the country
The Country mean CMD severity was moderate with mean CMD severity of 3.0. The lowest mean CMD severity (2.4) was observed in Southern Region closely followed by Northern region which recorded a mean severity of 2.9 and the highest CMD severity was recorded in the Western area region (3.7) according to the regional level assessment. The district level assessment revealed indicate that Bo (2.4) and Moyamba (2.8) districts in the Southern region and Bombali district (2.8) in the Northern region recorded low CMD severity whilst the highest CMD was observed in Western area urban (4.0) in the Western area region followed by severity 3.7 observed in Falaba district of the Northern region. The low to severity observed in Bo, Moyamba and Bombali can be attributed to the increase in the adoption and cultivation of improved cassava variety especially the SLICASS 4 that majority of the fields visited fall within CMD of 2-3 which is low to moderate CMD symptoms. The distribution map showed the widespread of CMD severity with many fields having a CMD of 2-3 (Figure 5). Fields with no CMD symptom or asymptomatic (healthy) were mostly found in few districts in South and northern regions.
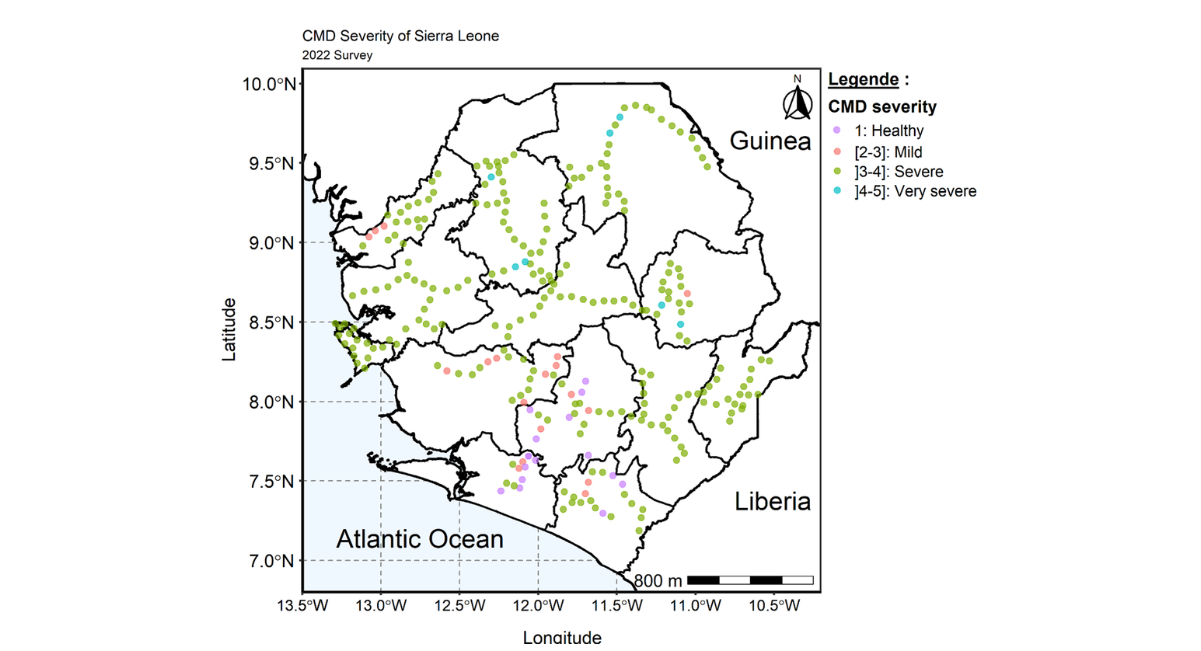
Figure 5: Map of Sierra Leone showing the distribution and spread of CMD severity across the country
The CMD symptom assessment at regional level revealed that in the eastern region, 140 farms out of the 278 visited were exhibiting mosaic pattern symptom, 66 farms were exhibiting leaf distortion, 37 exhibited veins clearing, whilst 32 of the fields exhibited chlorotic blotch (Table 4). Similarly, in the Northwest region, 132 fields out of the 278 assessed showed mosaic pattern symptom, 61 fields showed leaf distortion symptoms, whilst 34 of the fields exhibited chlorotic blotch and only 2 fields showed filiform like symptom. Similar observation was also done in the Southern region where 142 fields out of 278 showed mosaic like pattern symptom, 70 showed leaf distortion, whereas 26 showed chlorotic blotch and only 4 fields showed filiform symptom of CMD. In the Western area, it was revealed that 147 out of 278 fields visited showed mosaic like pattern symptom, 67 showed leaf distortion, whilst 39 showed chlorotic blotch and 3 fields showed filiform symptom.
Table 4: Cassava mosaic disease symptom assessment across regions of Sierra Leone
|
Symptoms |
|||||
|
Region |
Chlorotic blotch |
Filiform |
Leaf distortion |
Mosaic |
Vein clearing |
|
Eastern |
32 |
3 |
66 |
140 |
37 |
|
Northwest |
34 |
2 |
61 |
132 |
49 |
|
Southern |
26 |
4 |
70 |
142 |
36 |
|
Western |
39 |
3 |
67 |
147 |
22 |
Cassava samples in which only ACMV was detected and those in which the virus occurred in single infection as well as in mixed infections with EACMV predominated compared to EACMCMV (Table 5). Few cassava leaf samples contained EACMV in single infection in addition to mixed infections by ACMV and EACMV. The ACMV and EACMV were detected singly in 315 and 213 samples, respectively.
Table 5: Cassava mosaic disease symptoms on cassava associated with single and mixed infections by African cassava mosaic virus (ACMV) and East African cassava mosaic virus (EACMV)
|
Single |
Mixed |
Combined |
|||
|
Parameter |
ACMV |
EACMV |
ACMV+EACMV |
ACMV+EACMCMV |
|
|
Number of plants |
315 |
213 |
345 |
182 |
1055 |
|
Percent infected |
29.86 |
20.19 |
32.7 |
17.25 |
100 |
Whitefly population
The survey conducted revealed the country mean whitefly of 5.17 which was higher than the mean whitefly recorded during the 2020 national survey. Variation was observed in whitefly population in all survey sites across the country. The reginal level assessment indicate that whitefly counts varies from 4.03 in the southern region to 6.43 in the Western area region which also recorded the highest number of whitefly (1102.0). However, the Southern region recorded the lowest number of whitefly (242.0) (Table 6). The distribution map indicate whitefly are widely distributed across all regions and districts. The map also revealed that most of the fields assessed have whitefly counts above 6 (Figures 6 and 7).
|
Region |
Mean whitefly |
Total number of whiteflies |
Maximum |
Minimum |
Field surveyed |
|
Eastern |
4.4 |
402 |
11.34 |
0 |
60 |
|
Southern |
4.03 |
242 |
10 |
0 |
75 |
|
Northern |
4.88 |
579 |
16 |
0 |
75 |
|
Northwest |
6.12 |
1025 |
12 |
0 |
55 |
|
Western Area |
6.43 |
1102 |
15 |
0 |
13 |
|
Mean/sum |
5.17 |
3350 |
12.86 |
0 |
278 |
Table 6: Whitefly numbers observed in surveyed fields per region
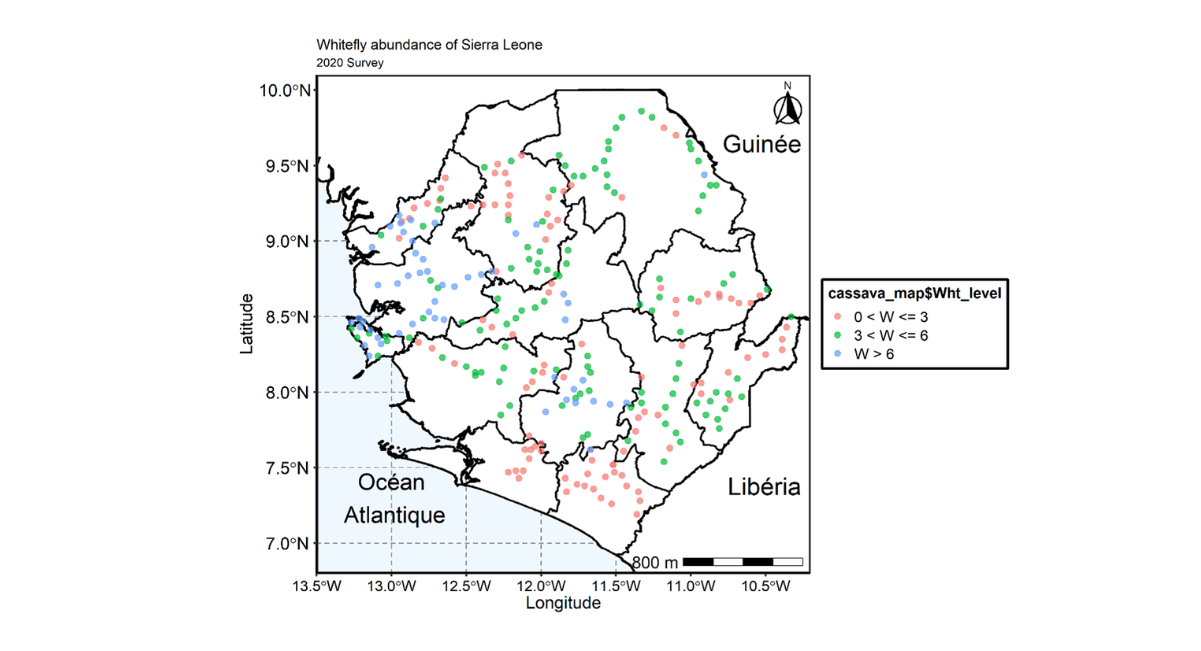
Figure 6: Map of Sierra Leone showing the distribution and spread of whitefly across the country during 2020 cropping season
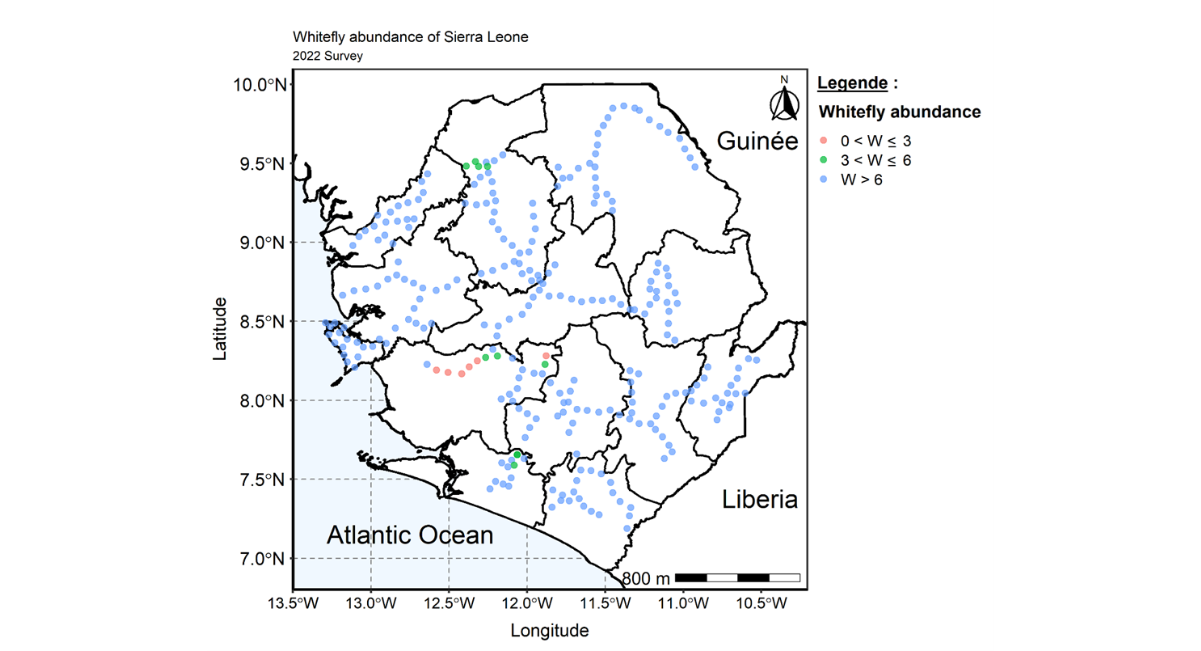
Figure 7: Map of Sierra Leone showing the distribution and spread of whitefly across the country during 2022 cropping season
Whitefly population was highest in fields with CMD incidence of >50-75% (5.05) which was closely followed with fields with CMD incidence of >75-100% (4.06) and healthy fields recorded the lowest whitefly population (1.02). The assessment revealed that as the CMD incidence increases so the number of whiteflies increases (Figure 8).
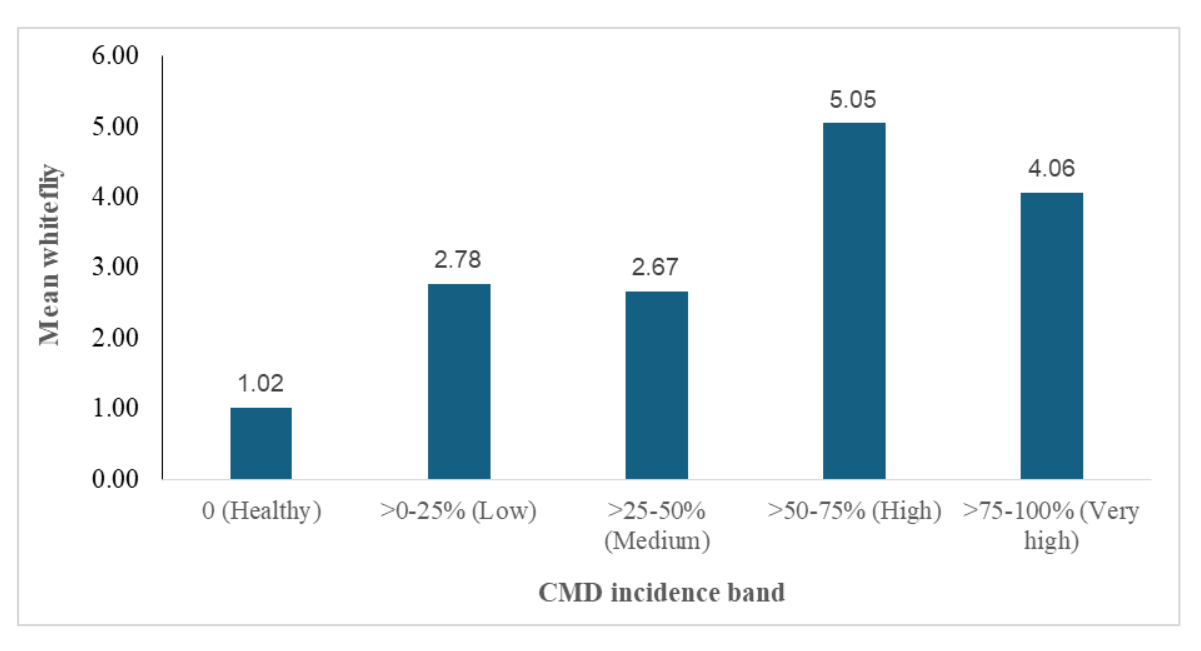
Figure 8: Mean whitefly by CMD incidence
Whitefly population by CMD Severity
The highest whitefly mean count (9.9) was counted on asymptomatic plants, whereas the highest CMD severity of 5 recorded the whitefly counts of 9.6 which was higher than what was recorded during 2020 assessment (Figure 9).
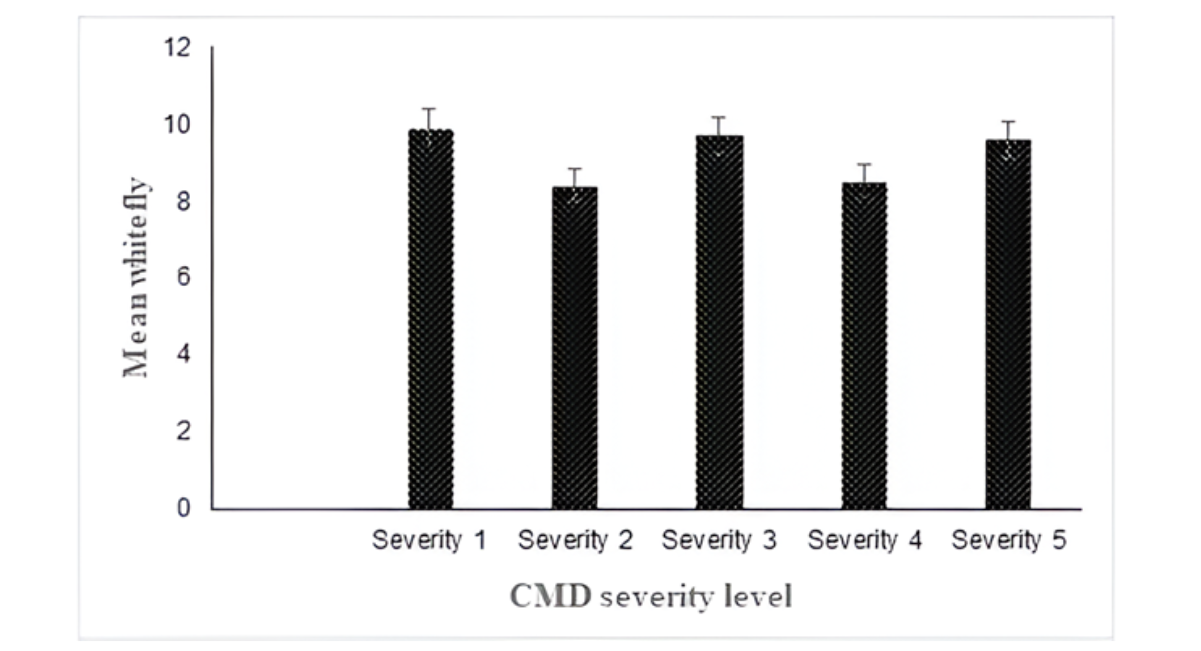
Figure 9: Mean whitefly by CMD severity
A statistically significant positive correlation (P<0.01) was observed between the incidence and severity of CMD, while a significant negative correlation was found between CMD incidence and whitefly population (Table 7). Additionally, there was a negative correlation between CMD severity and whitefly count. The result demonstrated a strong significant positive correlation (P < 0 .01) between altitude and whitefly abundance, indicating that lower altitude is associated with higher whitefly population.
Table 7: Pairwise Spearman rank correlation coefficients rs (normal text) and P-values (italics)
|
Parameters |
CMD |
CMD |
Whitefly |
Altitude |
Plant Age |
|
Incidence |
Severity |
Abundance |
|||
|
CMD Incidence |
1 |
0.54** |
-0.21* |
0.20* |
0.35* |
|
0.001 |
0.015 |
0.03 |
0.002 |
||
|
CMD Severity |
1 |
-0.14 |
-0.52** |
0.56** |
|
|
0.234 |
0.001 |
0.001 |
|||
|
Whitefly abundance |
1 |
0.43** |
0.16 |
||
|
0.001 |
0.36 |
||||
|
Altitude |
1 |
0.12 |
|||
|
0.2 |
|||||
|
Plant Age |
1 |
**, significant at P < 0.01 and *, significant at P < 0.05 (two-tailed), n = 278.
Identification of new Alternate Hosts of ACMV and EACMV
The ACMV and EACMV co-infected a leguminous weed, Senna occidentalis (L.) Link, Carica papaya, (b) Centrosema pubescens, and (c) Manihot glaziovii (wild cassava) showing mosaic symptoms (Figure 10) as diagnosed by PCR (Figure 11).

Figure 10: Images of some alternate hosts of cassava mosaic disease (CMD) identified in (a) Carica papaya, (b) Centrosema pubescens, and (c) Manihot glaziovii (wild cassava)
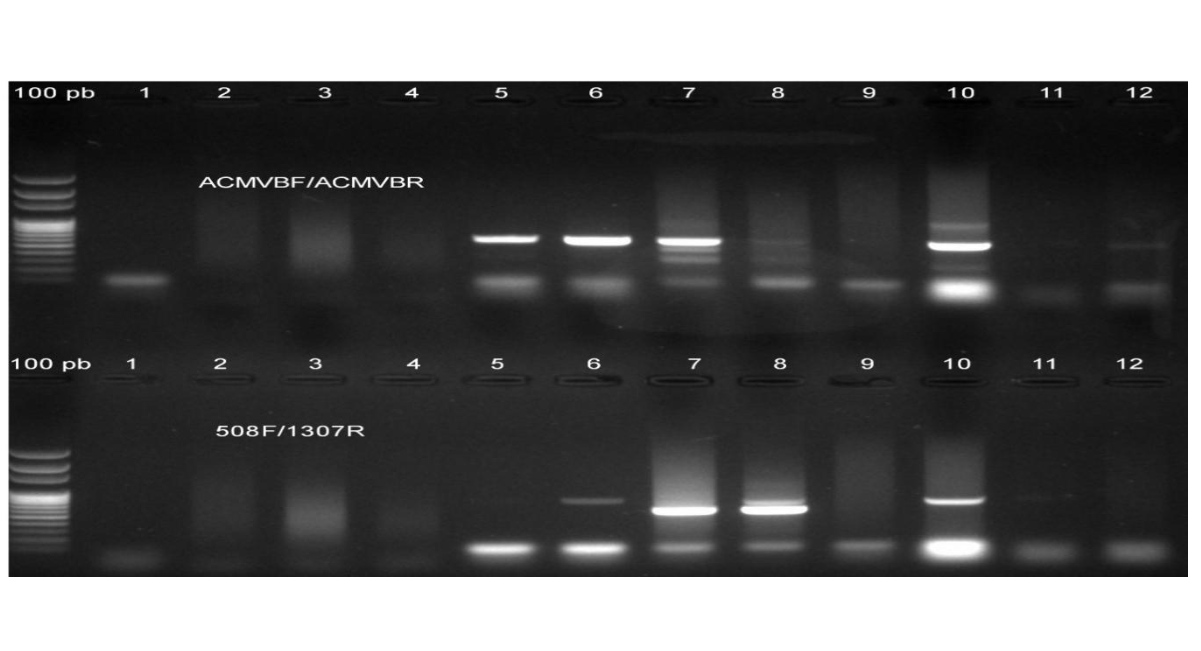
Figure 11: Gel electrophoresis of DNA fragments of African cassava mosaic virus (ACMV) and East African cassava mosaic virus (EACMV) in weed samples in cassava fields in Sierra Leone amplified in polymerase chain reaction by two pairs of primers. Lanes 1–3 contained total DNA of weed samples Leucaena feucocephafa, Ageratum conyzoides (L.), and Sida spp., respectively; lanes 4–5 contained DNA of Centrosema pubescens; lane 6 contained DNA of Senna occidentalis (L.); lanes 7-8 contained DNA of Carica papaya; lanes 9-10 = DNA of wild cassava (Manihot glaziovii Muller von Argau) leaf co-infected by ACMV and EACMV as positive control. The samples were tested by primers: ACMVBVF/ACMVBVR and 508F/1307R designed for detection of ACMV and EACMV, respectively of the 10 symptomatic alternate host leaf samples analysed, primer ACMVBF/ACMVBR detected four symptomatic samples of ACMV, representing 40% (4/10), and primer WAVE-508F/WAVE-1307R also detected four samples for EACMV-like virus, representing 40% (4/10), while the remaining six samples were negative, representing 60% (6/10). The four alternate hosts detected at the molecularly level with begomoviruses include Senna occidentalis, Centrosema pubescens, Carica papaya and Manihot glaziovii.
DISCUSSION
The predominance of single infections of ACMV over single infections of EACMV in Sierra Leone had been reported from a survey conducted in 2020 [27]. In that survey, ACMV-like virus was diagnosed in 17.85% (40/224) in a single infection out of the 224 samples tested. Findings of previous studies are similar to the present survey in which 315 (29.86%), and 213 (20.19%) samples yielded single infections of ACMV and EACMV, respectively, out of the 1055 samples tested. Unlike Tanzania, Kenya, Malawi and Madagascar, where single infections by EACMV were predominant [28–31], in Cote d’Ivoire [24] and Cameroon [32], single infections were lower than mixed infections. More and wider distribution of mixed infections by ACMV+EACMV and ACMV+EACMCMV were recorded in the present survey than in the survey conducted in 2020. The 2020 survey found 14.73% (ACMV+EACMV) and 8.93% (ACMV+EACMCMV) mixed infections compared with the 32.70% (ACMV+EACMV) and 17.25% (ACMV+EACMCMV) in the present survey. More routes were covered, more farms were visited, and more plants per farm were sampled than in the 2020 to increase the sensitivity of the present survey, particularly with respect to detecting EACMV and EACMCMV. The intensive cultivation of cassava crop could have possibly contributed to the indiscriminate movement of infected planting materials, and the dissemination of the ACMV and EACMV by whitefly vectors, leading to a greater number of farms with mixed infections.
The high prevalence of CMD across the country due to the farmers’ preference of cultivating more of the local varieties that are susceptible to the disease, but possess poundablity or ability to cook well, sweetness and high dry matter content. Similarly, in Ghana, Oppong et al. [33] found that farmers were predominantly cultivating local varieties that are susceptible to the CMD. Accordingly, most of the farmers in Sierra Leone are not aware of the CMD and its associated vectors, as they often associate cassava mosaic symptoms on young leaves as being having too much nutrient and therrefore are good for cooking because they believe that young leaves with mosaic symptoms are good for sweet cassava leaf sauce preparation. This implies that most farmers in the country do not perceive CMD as a significant threat for cassava production due to ignorance of the disease, and the devastating damage it causes; as such, they often neglect good management practices that mitigate the devastating effects of the disease. These findings are reminiscent of those obsereved by Chikoti et al. [34] and Samura et al. [35] that strong cultural attachment of farmers influence the predominant cultivation of local varieties. Farmers in Sierra Leone favor varieties that possess low cyanide content, high dry matter content, and poundable (easy to cook), especially for production targeting the fresh product market; and they are not too particular about the yield of the cassava. Farming women opined that the low chlorophyll content in the mosaic-infected leaves reduces the amount of palm oil required for cooking compared to the disease-free improved cassava genotypes. Palm oil is an expensive yet crucial ingredient in preparing cassava leaf sauce in Sierra Leone. It was observed from the assessment that most of the CMD symptoms assessed on plants were indicative of cutting-borne infection than whitefly derived infection, this situation can be attributed to the farm-to-farm exchange of planting materials/cuttings as a source of acquiring plantings within the farming communities.
The significant positive correlation between CMD incidence and severity (r = 0.54**) and a significant negative CMD incidence and whitefly abundance (r = -0.21*) obtained in this study are similar to findings of Samura et al. [27], who noted positive (r=0.423**) and negative (r = -0.530**) associations, respectively. The negative correlation between whitefly presence and CMD incidence and between whitefly abundance and severity suggests that the CMD symptom observed are possibly attributable to cutting-borne which suggest whitefly presence or absence does not significantly affect the CMD incidence and severity levels. Eni et al. [36] also found no correlation between whitefly number and CMD incidence. Accordingly, in the Western area region, despite the high whitefly population found in the small farms, a moderate CMD severity of the disease was detected. Moreover, plants with high CMD infection and low whitefly population suggest that whitefly is more attracted to plants with less CMD symptom partly due to their unpalatability for fecundation. This agrees with the view of Wamonje et al. [37], who reported that virus infected cassava leaves create a repellent or unattractive environment for whitefly to settle.
Most of the plants assessed exhibited symptoms of CMD that are associated with cutting borne infection with CMD symptoms found both on upper and lower leaves and the most predominant CMD symptoms were mosaic and leaf distortion. Legg et al. [38] reported that cassava mosaic virus is primarily transmitted through persistent infections from the whitefly Bemisia tabaci but is also widely propagated through infected stem cuttings. This observation was consistent with the results of this study which largely attributed CMD prevalence to the continuous cultivation of susceptible cassava cultivars and whiteflies. Our field observations revealed that resistant varieties, mainly used for commercial purposes, were also planted alongside local varieties as secondary options.
The assessment noted variation in whitefly population among farms in all regions across the country with an average whitefly of 5.17, attributable to environmental conditions such as climate variability, whitefly biotype, altitude, cassava varieties and agronomic practices. The average whitefly value obtained in this study was lower than those recorded in Cameroon [39], but higher than the whitefly population reported in Burkina Faso [20] and in Ghana [33]. The assessment further revealed regional differences in whitefly abundance with Western area region exhibiting higher whitefly abundance followed by Northwest region despite recording low number of farms. The abundance of whitefly in the Western area and Northwest regions maybe as the result of the proximity of these regions to the Atlantic Ocean with lower altitude and higher humidity which favor whitely settlement. Similarly, the observation made in Madagascar by Harimalala et al. [40] and in Tanzania by Szynizewska et al. [41], where whitefly counts decreased with higher altitude were consistent with this study. Although previous studies [42] have shown a clear correlation between whitefly abundance and the transmission of whitefly-borne infections, it is challenging to establish a direct causal relationship due to temporal variations in whitefly populations [43] and the latency period of 3 to 5 weeks between transmission and the appearance of the first symptoms [44]. In the Eastern and Northern regions, whitefly population was low, but CMD incidence and severity were high and moderate, respectively. Similar observations were made by Houngue et al. [45] in the Alibori and Borgou regions of Benin, where they reported low whitefly populations despite high or medium CMD incidence. This indicates that whiteflies do not act only as vectors for CMD viruses but contribute to the spread of other viruses [46]. The active presence of whiteflies and the high incidence and severity of cassava mosaic disease in Sierra Leone significantly impact the disease's epidemiology across the Mano River Union countries and the Economic Community of West African States (ECOWAS) region. Research in Sierra Leone suggests that cassava production will remain low and uncompetitive, posing challenges for the emerging small and medium-scale industries in the cassava subsector, such as gari and flour enterprises [7, 47].
The detection of ACMV and EACMV in Senna occidentalis, Centrosema pubescens, Carica papaya and Manihot glaziovii is a novel finding in Sierra Leone. Ogbe et al. [48] identified Nicotiana benthamiana Domin., as an experimental host of ACMV and EACMV. Shoyinka et al. [49] had reported castor oil plant (R. communis) as a natural host of ACMV and EACMV. The negative results obtained in weed sample isolates for the diagnosis of ACMV and EACMV indicate that these viruses might not be present in them. However, there is need of further characterization of more putative weed samples with CMD using more efficient markers that could detect strains of cassava mosaic begomoviruses existing in the country. This supports the view that genetic modification by mutation or recombination of geminiviruses has produced new species and strains including EACMV-Ug2 [50] and SACMV [51] that could be exploited.
CONCLUSION
This study establishes the prevalence of cassava mosaic disease (CMD) across all regions of Sierra Leone, with evidence of its continuous spread into new cassava-growing areas. In some districts, CMD prevalence has reached 100%, posing a significant threat of high yield losses due to the severe mosaic symptoms observed in most fields. The recorded incidence and severity of CMD are alarming, with high whitefly populations than those recorded in 2020 survey. The exchange of contaminated planting materials among farmers creates optimal conditions for the introduction and spread of virulent cassava mosaic geminivirus (CMG) species or strains, potentially leading to a severe CMD pandemic. To effectively manage CMD and enhance cassava productivity, an integrated strategy is essential. This strategy should include the multiplication, distribution, and adoption of improved cassava planting materials that are resistant or tolerant to CMD for local farmers. Additionally, training farmers to recognize CMD and promoting the use of healthy cuttings when establishing new plots are crucial steps. Future efforts should focus on characterizing the viruses and vectors associated with CMD, strengthening phytosanitary and quarantine measures, and implementing frequent surveillance or monitoring programs. These measures will help prevent further CMD spread and minimize its impact, thereby mitigating the potential for widespread outbreaks.
Credit authorship contribution statement
Musa Decius Saffa: Conceptualization; Data curation; Methodology; Investigation; Formal analysis; Writing-original draft.
Prince Emmanuel Norman: Visualization; Validation; Formal analysis; Writing-review & editing.
Adama Sagnon: Investigation; Methodology; Data curation; Validation; Formal analysis; Software.
Kadiatu Serry: Investigation; Writing review and editing.
All authors have read and agreed to the published version of the manuscript.
ACKNOWLEDGMENTS
This work was supported by the Bill and Melinda Gates Foundation and The United Kingdom Foreign, Commonwealth and Development Office (FCDO) under a grant with the investment record ID OPP1212988/INV-002969 to the Central and West African Virus Epidemiology (WAVE) Program for root and tuber crops through a subgrant from the Université Félix Houphouët-Boigny (UFHB) to Njala University. The authors thank the crop protection and cassava breeding team at Njala University, the Sierra Leone Agricultural Research Institute (SLARI), and WAVE Ivory Coast for their technical support during this study.
DATA AVAILABILITY
The corresponding author can make data from this research available upon request.
DECLARATION OF COMPETING INTEREST
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
REFERENCES
- Maziya-Dixon B, Dixon A G O, Adebowale A A, et al. (2019). Cassava: Africa’s food security crop in the face of climate change. Food Security, 11: 1083–1098.
- Akinola A, Ojo B, Adeyemi C. (2020). Resilience and superior yield performance of cassava compared to other root and tuber crops. Journal of Crop Science, 25: 215–224.
- Ufuan Achidi A, Ajayi OA, Bokanga M, Maziya-Dixon B. The use of cassava leaves as food in Africa. Ecol. Food Nutr. 2005;44(6):423-35. [Google Scholar] [Crossref]
- Devi B, Kumar MN, Chutia M, Bhattacharyya N. Abiotic and biotic stress challenges of cassava in changing climate and strategies to overcome: a review. Sci. Hortic. 2022;305:111432. [Google Scholar] [Crossref]
- Amara V, Samura AE, Norman PE, Kanu SA, Karim KY, et al. Characterization of the Cassava (Manihot esculenta) germplasm in Sierra Leone Based on agro-morphological and culinary traits. Horticulturae. 2024;10(6):640. [Google Scholar] [Crossref]
- FAOSTAT (2015). Food and Agriculture Organization of the United Nations. FAOSTAT data base. Available at [Google Scholar] [Crossref]
- Samura AE, Lakoh KA, Nabay O, Fomba SN, Koroma JP. Effect of cassava mosaic disease (CMD) on yield and profitability of cassava and gari production enterprises in Sierra Leone. J. Agric. Sci. 2017;9(205):10-5539. [Google Scholar] [Crossref]
- Bhaargavi R, Latha TK, Makeshkumar T, Harish S. Cassava mosaic disease: strategies for recovery and sustainable management. Australas. Plant Pathol. 2025;54(1):1-2. [Google Scholar] [Crossref]
- Bull SE, Briddon RW, Sserubombwe WS, Ngugi K, Markham PG, et al. Genetic diversity and phylogeography of cassava mosaic viruses in Kenya. J Gen Virol. 2006;87(10):3053-65. [Google Scholar] [PubMed] [Crossref]
- Berrie LC, Rybicki EP, Rey ME. Complete nucleotide sequence and host range of South African cassava mosaic virus: further evidence for recombination amongst begomoviruses. J Gen Virol. 2001;82(1):53-8. [Google Scholar] [PubMed] [Crossref]
- Legg JP, Owor B, Sseruwagi P, Ndunguru J. Cassava mosaic virus disease in East and Central Africa: epidemiology and management of a regional pandemic. Adv Virus Res. 2006;67:355-418. [Google Scholar] [PubMed] [Crossref]
- Smart O, Vaikosen SE, Bamgbose AM. Nutrient composition of cassava offals and cassava sievates collected from locations in Edo state, Nigeria. Pak. J. Nutr. 2005;4(4):262-4. [Google Scholar] [Crossref]
- Samura AE, Massaquoi FB, Mansaray A, Kumar PL, Koroma JP, et al. Status and diversity of the cassava mosaic disease causal agents in Sierra Leone. Int. J. Agric. For. 2014;4(3):246-54. [Google Scholar]
- Samura P, Kamara A, Moses C, et al. Farmer participatory selection of cassava varieties in Sierra Leone. International J. Agron. Agric. Res. 2014; 5(5): 61–72.
- Fauquet CM, Bisaro DM, Briddon RW, Brown JK, Harrison BD, et al. Revision of taxonomic criteria for species demarcation in the family Geminiviridae, and an updated list of begomovirus species. Arch Virol. 2003;148(2):405-20. [Google Scholar] [PubMed] [Crossref]
- Thresh J M, Otim-Nape G W, Legg J P, et al. (1997). Africa cassava mosaic disease: what is the magnitude of the problem? African Journal of Root and Tuber Crops Special Issue: Contributions of Biotechnology to Cassava in Africa. Proceedings of the Cassava Biotechnology Network, Third International Scientific Meeting. Kampala, Uganda 26-31 August 1996. Eds Thro RM, Akoroda MP pp. 13–19.
- Samura AE, Kanteh SM, Norman JE, Fomba SN. Integrated pest management options for the cassava mosaic disease in Sierra Leone. Int. J. Agric. Innov. Res. 2016;5(3):432-40. [Google Scholar]
- Janatu VS, Kwadwo OA, Prince EN. Shoot nodal culture and virus indexing of selected local and improved genotypes of cassava (Manihot esculenta) from Sierra Leone. Int. J. Biotechnol. Mol. Biol. Res. 2016;7(2):20-8. [Google Scholar] [Crossref]
- Eni AO, Efekemo OP, Onile‐ere OA, Pita JS. South West and North Central Nigeria: Assessment of cassava mosaic disease and field status of African cassava mosaic virus and East African cassava mosaic virus. Ann Appl Biol. 2021;178(3):466-79. [Google Scholar] [PubMed] [Crossref]
- Soro M, Tiendrébéogo F, Pita JS, Traoré ET, Somé K, et al. Epidemiological assessment of cassava mosaic disease in Burkina Faso. Plant Pathol. 2021;70(9):2207-16. [Google Scholar] [PubMed] [Crossref]
- Sseruwagi P, Sserubombwe WS, Legg JP, Ndunguru J, Thresh JM. Methods of surveying the incidence and severity of cassava mosaic disease and whitefly vector populations on cassava in Africa: a review. Virus Res. 2004;100(1):129-42. [Google Scholar] [PubMed] [Crossref]
- International Institute of Tropical Agriculture. Cassava in tropical Africa: a reference manual. IITA; 1990. [Google Scholar]
- Andreola S, Rodriguez M, Parola R, Alemano S, Lascano R. Interactions between soybean, Bradyrhizobium japonicum and Soybean mosaic virus: the effects depend on the interaction sequence. Funct Plant Biol. 2019;46(11):1036-48. [Google Scholar] [PubMed] [Crossref]
- Pita JS, Fondong VN, Sangaré A, Kokora RN, Fauquet CM. Genomic and biological diversity of the African cassava geminiviruses. Euphytica. 2001;120(1):115-25. [Google Scholar] [Crossref]
- Matic S, Da Cunha AP, Thompson JR, Tepfer M. An analysis of viruses associated with cassava mosaic disease in three Angolan provinces. J. Plant Pathol. 2012:443-50. [Google Scholar]
- Alabi OJ, Kumar PL, Naidu RA. Multiplex PCR for the detection of African cassava mosaic virus and East African cassava mosaic Cameroon virus in cassava. J Virol Methods. 2008;154(1-2):111-20. [Google Scholar] [PubMed] [Crossref]
- Samura AE, Amara V, Bah AM, Johnson R, Saffa MD. Cassava Mosaic Disease CMD and Its Whitefly Vector in Sierra Leone: Incidence, Severity and Whitefly Population from Field Surveys in 2020. Adv. Plant Pathol. Res. 2025;1(1):16-25. [Google Scholar]
- Ogbe FO, Dixon AG, Hughes JD, Alabi OJ, Okechukwu R. Status of cassava begomoviruses and their new natural hosts in Nigeria. Plant Disease. 2006;90(5):548-53. [Google Scholar] [PubMed] [Crossref]
- Ogbe FO, Legg J, Raya MD, Muimba-Kankolongo A, Theu MP, et al. Diagnostic survey of cassava mosaic viruses in Tanzania, Malawi and Zambia. Roots. 1997;4(2):12-5. [Google Scholar]
- Ogbe FO, Thottappilly G, Quin FM. Implementation in Africa of serological diagnostic test for cassava mosaic geminiviruses. Afr. J. Root Tuber Crops. 1997;2(1/2):33-6. [Google Scholar]
- Swanson M M, Harrison B D. Properties, relationships and distribution of cassava mosaic geminiviruses. Tropical Science. 1994;34(1): 15–25. [Google Scholar]
- Fondong VN, Pita JS, Rey ME, De Kochko A, Beachy RN, et al. Evidence of synergism between African cassava mosaic virus and a new double-recombinant geminivirus infecting cassava in Cameroon. J Gen Virol. 2000;81(1):287-97. [Google Scholar] [PubMed] [Crossref]
- Oppong A, Prempeh RN, Abrokwah LA, Annang EA, Marfo EA, et al. Cassava mosaic virus disease in Ghana: distribution and spread. [Google Scholar] [Crossref]
- Chikoti PC, Melis R, Shanahan P. Farmer’s perception of cassava mosaic disease, preferences and constraints in Lupaula Province of Zambia. Am. J. Plant Sci. 2016;7(07):1129. [Google Scholar] [Crossref]
- Samura AE, Fomba SN, Mansarsy A, Saffa MD, Quee DD, et al. Seasonal Variation on the Incidence and Severity of Major Foliar Diseases of Cassava in Sierra Leone. InCassava-Biology. 2021. IntechOpen. [Google Scholar] [Crossref]
- Eni AO, Efekemo OP, Soluade MG, Popoola SI, Atayero AA. Incidence of cassava mosaic disease and associated whitefly vectors in South West and North Central Nigeria: Data exploration. Data Brief. 2018;19:370-92. [Google Scholar] [PubMed] [Crossref]
- Wamonje FO, Tungadi TD, Murphy AM, Pate AE, Woodcock C, et al. Three aphid-transmitted viruses encourage vector migration from infected common bean (Phaseolus vulgaris) plants through a combination of volatile and surface cues. Frontiers in Plant Science. 2020;11:613772. [Google Scholar] [PubMed] [Crossref]
- Legg J P, Jeremiah S C, Obiero H M, et al. (2021). Diversity and distribution of cassava mosaic begomoviruses associated with cassava mosaic disease in Africa and the Indian subcontinent. Virus Research, 200(5): 98–107. [Google Scholar] [PubMed] [Crossref]
- Doungous O, Masky B, Levai DL, Bahoya JA, Minyaka E, et al. Cassava mosaic disease and its whitefly vector in Cameroon: Incidence, severity and whitefly numbers from field surveys. Crop Prot. 2022;158:106017. [Google Scholar] [PubMed] [Crossref]
- Harimalala M, Chiroleu F, Giraud‐Carrier C, Hoareau M, Zinga I, et al. Molecular epidemiology of cassava mosaic disease in M adagascar. Plant Pathology. 2015;64(3):501-7. [Google Scholar] [PubMed]
- Szyniszewska AM, Busungu C, Boni SB, Shirima R, Bouwmeester H, et al. Spatial analysis of temporal changes in the pandemic of severe cassava mosaic disease in northwestern Tanzania. Phytopathology. 2017;107(10):1229-42. [Google Scholar] [PubMed] [Crossref]
- Legg JP, Raya MD. Survey of cassava virus diseases in Tanzania. Int. J. Pest Manag. 1998;44(1):17-23. [Google Scholar]
- Fishpool LD, Fauquet C, Fargette D, Thouvenel JC, Burban C, et al. The phenology of Bemisia tabaci (Homoptera: Aleyrodidae) populations on cassava in southern Côte d'Ivoire. Bull. Entomol. Res.. 1995;85(2):197-207. [Google Scholar] [Crossref]
- Fargette D, Jeger M, Fauquet C, Fishpool LD. Analysis of temporal disease progress of African cassava mosaic virus. Phytopathology. 1994;84(1):91-8. [Google Scholar] [Crossref]
- Houngue JA, Houédjissin SS, Ahanhanzo C, Pita JS, Houndénoukon MS, et al. Cassava mosaic disease (CMD) in Benin: Incidence, severity and its whitefly abundance from field surveys in 2020. Crop Protection. 2022;158:106007. [Google Scholar] [PubMed] [Crossref]
- Chikoti PC, Ndunguru J, Melis R, Tairo F, Shanahan P, et al. Cassava mosaic disease and associated viruses in Zambia: occurrence and distribution. Int. J. Pest Manag.. 2013;59(1):63-72. [Google Scholar] [Crossref]
- Nabay O, Conteh A R, Samura A E, et al.. Economic analysis of gari processing systems in Sierra Leone. Asian J. Agric. Ext. Econ. Sociol. 2018;26(3): 1–10. [Google Scholar]
- Ogbe FO, Atiri GI, Dixon AG, Thottappilly G. Serological and biological variations of African cassava mosaic virus, in Nigeria. Ann. Appl. Biol. 2003;143(2):203-13. [Google Scholar] [Crossref]
- Shoyinka S A, Thottappilly G, McGrath F F, Harrison B D. (2001). Detection, relationships and properties of cassava mosaic geminivirus in naturally infected castor oil plant, Ricinus communis L. in Nigeria. Page 20 in: The Book of Abstracts, Fifth Conf. Cassava Biotech. Network (CBN-V), Donald Danforth Plant Science Center, St. Louis.
- Zhou X, Liu Y, Calvert L, Munoz C, Otim-Nape GW, et al. Evidence that DNA-A of a geminivirus associated with severe cassava mosaic disease in Uganda has arisen by interspecific recombination. J Gen Virol. 1997;78(8):2101-11. [Google Scholar] [PubMed] [Crossref]
- Berrie LC, Rybicki EP, Rey ME. Complete nucleotide sequence and host range of South African cassava mosaic virus: further evidence for recombination amongst begomoviruses. J Gen Virol. 2001;82(1):53-8. [Google Scholar] [PubMed] [Crossref]